47132
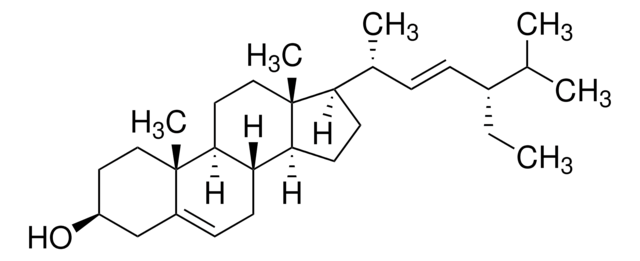
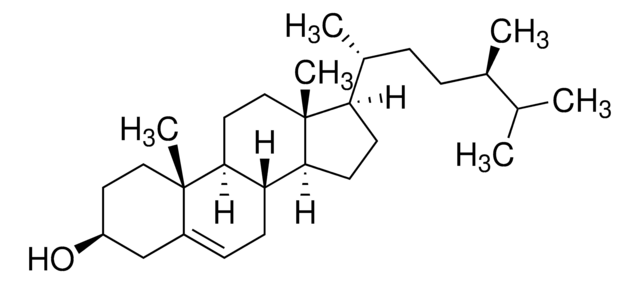
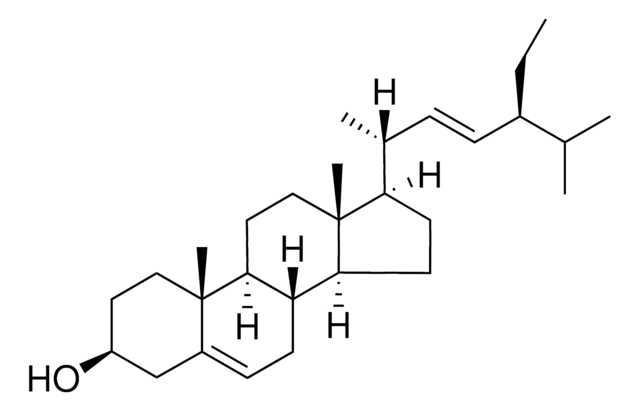
Stigmasterol
certified reference material, 10 mg/mL in chloroform
Synonym(s):
3β-Hydroxy-24-ethyl-5,22-cholestadiene, 5,22-Stigmastadien-3β-ol, Stigmasterin
About This Item
Recommended Products
grade
certified reference material
TraceCERT®
product line
TraceCERT®
Assay
95% (chromatography)
form
liquid
CofA
current certificate can be downloaded
packaging
ampule of 1 mL
concentration
10 mg/mL in chloroform
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
mp
165-167 °C (lit.)
application(s)
food and beverages
format
single component solution
functional group
hydroxyl
storage temp.
2-30°C
room temp
SMILES string
[H][C@@]12[C@]([C@](CC[C@H](O)C3)(C)C3=CC2)([H])CC[C@@]4(C)[C@@]1([H])CC[C@]4([H])[C@]([H])(C)/C=C/[C@@H](CC)C(C)C
InChI
1S/C29H48O/c1-7-21(19(2)3)9-8-20(4)25-12-13-26-24-11-10-22-18-23(30)14-16-28(22,5)27(24)15-17-29(25,26)6/h8-10,19-21,23-27,30H,7,11-18H2,1-6H3/b9-8+/t20-,21-,23+,24+,25-,26+,27+,28+,29-/m1/s1
InChI key
HCXVJBMSMIARIN-PHZDYDNGSA-N
Gene Information
human ... POLA1(5422) , TOP2A(7153)
rat ... Polb(29240)
Looking for similar products? Visit Product Comparison Guide
Application
Other Notes
Legal Information
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 3 - Carc. 2 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2 - STOT RE 1 - STOT SE 3
Target Organs
Central nervous system, Liver,Kidney
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Separation of Cholesterol; Brassicasterol; Campesterol; Stigmasterol; β-Sitosterol
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service