411205
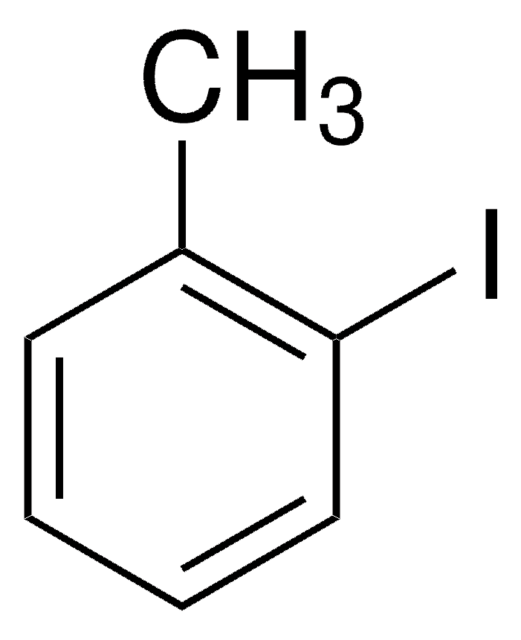
4-tert-Butyliodobenzene
98%
Synonym(s):
1-tert-Butyl-4-iodobenzene
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(CH3)3CC6H4I
CAS Number:
Molecular Weight:
260.11
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
liquid
refractive index
n20/D 1.57 (lit.)
bp
116-118 °C/9 mmHg (lit.)
density
1.468 g/mL at 25 °C (lit.)
SMILES string
CC(C)(C)c1ccc(I)cc1
InChI
1S/C10H13I/c1-10(2,3)8-4-6-9(11)7-5-8/h4-7H,1-3H3
InChI key
WQVIVQDHNKQWTM-UHFFFAOYSA-N
Related Categories
General description
4-tert-Butyliodobenzene is an electron-rich aryl iodide. Heck reaction between 2-methylprop-2-en-1-ol and 4-tert-butyliodobenzene catalyzed by ionic liquids has been studied. It participates in the one-pot Heck-reductive amination reaction pathway during the synthesis of fungicide fenpropimorph.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Functionalised ionic liquids: synthesis of ionic liquids with tethered basic groups and their use in Heck and Knoevenagel reactions.
Forsyth SA, et al.
New. J. Chem., 34(4), 723-731 (2010)
Jungwoon Kim et al.
Molecules (Basel, Switzerland), 23(4) (2018-04-13)
Solution-processed organic light-emitting diodes (OLEDs) are attractive due to their low-cost, large area displays, and lighting features. Small molecules as well as polymers can be used as host materials within the solution-processed emitting layer. Herein, we report two 3,3'-bicarbazole-based host
One-pot multistep synthetic strategies for the production of fenpropimorph using an ionic liquid solvent.
Forsyth SA, et al.
Organic Process Research & Development, 10(1), 94-102 (2006)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service