All Photos(1)
About This Item
Empirical Formula (Hill Notation):
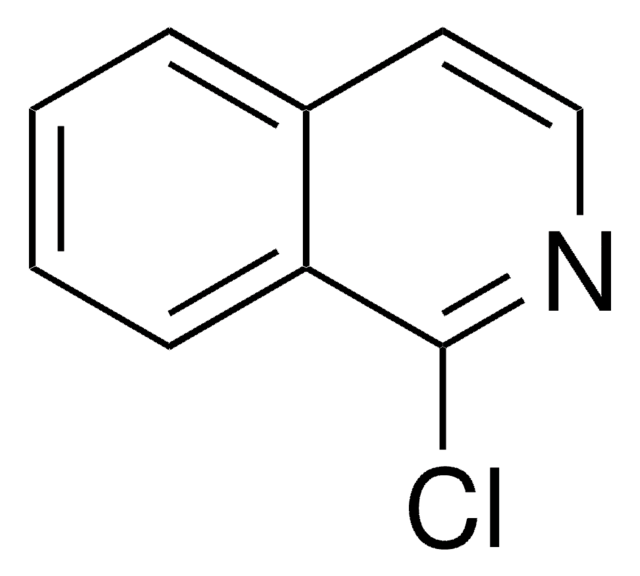
C9H5Cl2N
CAS Number:
Molecular Weight:
198.05
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
121-122 °C (lit.)
SMILES string
Clc1cc2ccccc2c(Cl)n1
InChI
1S/C9H5Cl2N/c10-8-5-6-3-1-2-4-7(6)9(11)12-8/h1-5H
InChI key
BRGZEQXWZWBPJH-UHFFFAOYSA-N
Related Categories
General description
1,3-Dichloroisoquinoline undergoes Pd(PPh3)4 catalyzed regioselective coupling with arylboronic acids to afford to 1-aryl-3-chloroisoquinolines. Reaction of amine with 1,3-dichloroisoquinoline has been studied. Regioselectivity of the Stille coupling reaction of (1-ethoxyvinyl)tri(n-butyl)stannane with 1,3-dichloroisoquinoline has been investigated.
Application
1,3-Dichloroisoquinoline may be used in the facile synthesis of 1,3,4-trisubstituted isoquinoline derivatives.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Syntheses of acetylquinolines and acetylisoquinolines via palladium-catalyzed coupling reactions.
Legros J-Y, et al.
Tetrahedron, 57(13), 2507-2514 (2001)
A facile synthesis of 1, 3, 4-trisubstituted isoquinolines.
Yang H.
Tetrahedron Letters, 50(25), 3081-3083 (2009)
Synthetic antimalarials; some derivatives of phthalazine, quinoxaline, and isoquinoline.
H D HAWORTH et al.
Journal of the Chemical Society, 174, 777-782 (1948-06-01)
Exploitation of differential reactivity of the carbon?chlorine bonds in 1, 3-dichloroisoquinoline. Routes to new N, N-chelate ligands and 1, 3-disubstituted isoquinolines.
Ford A, et al.
Journal of the Chemical Society. Perkin Transactions 1, 6, 927-934 (1997)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service