추천 제품
일반 설명
4-Biphenylacetic acid is a potential non-steroidal antiinflammatory agent and forms solid inclusion complex with β-cyclodextrin. 4-Biphenylacetic acid on interaction with quinolone antibacterial agents induces functional blockade of the γ-aminobutyric acid receptors.
애플리케이션
4-Biphenylacetic acid was used in the synthesis of gastrosparing non-steroidal antiinflammatory drug.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
Yohei Sakaguchi et al.
Journal of pharmaceutical and biomedical analysis, 55(1), 176-180 (2011-02-12)
Fluorous derivatization followed by fluorous-phase liquid chromatographic (LC) separation exploits the affinity between perfluoroalkyl compounds for highly selective and quantitative isolation of various analytes. However, the applicability of this technique as a simple pretreatment for fluorometric determination in clinical settings
M Koutsoviti-Papadopoulou et al.
Pharmacological research, 44(3), 229-233 (2001-09-01)
This paper examines the effect of biphenylacetic acid on the antagonistic action of norfloxacin and enoxacin on the GABA(A)-mediated responses of the isolated guinea-pig ileum. GABA produced transient contractions followed by relaxation. The contractile effect of exogenously applied GABA was
A I Khan et al.
Chemical communications (Cambridge, England), (22)(22), 2342-2343 (2002-09-21)
A series of pharmaceutically active compounds including diclofenac, gemfibrozil, ibuprofen, naproxen, 2-propylpentanoic acid, 4-biphenylacetic acid and tolfenamic acid can be reversibly intercalated into a layered double hydroxide, initial studies suggest that these materials may have application as the basis of
G Puglisi et al.
The Journal of pharmacy and pharmacology, 43(6), 430-432 (1991-06-01)
4-Biphenylacetic acid, a potent non-steroidal anti-inflammatory agent forms a solid inclusion complex with beta-cyclodextrin in a 1:1 molar ratio, which exhibits better solubility and dissolution characteristics than the uncomplexed drug. Following oral administration of the complex to rats, quicker and
K Akahane et al.
Antimicrobial agents and chemotherapy, 38(10), 2323-2329 (1994-10-01)
The combination of some new quinolone antibacterial agents with 4-biphenylacetic acid (BPAA), a metabolite of fenbufen, is known to specifically induce functional blockade of the gamma-aminobutyric acid (GABA) receptors. The mechanisms of these drug interactions were further examined. Scatchard analysis
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.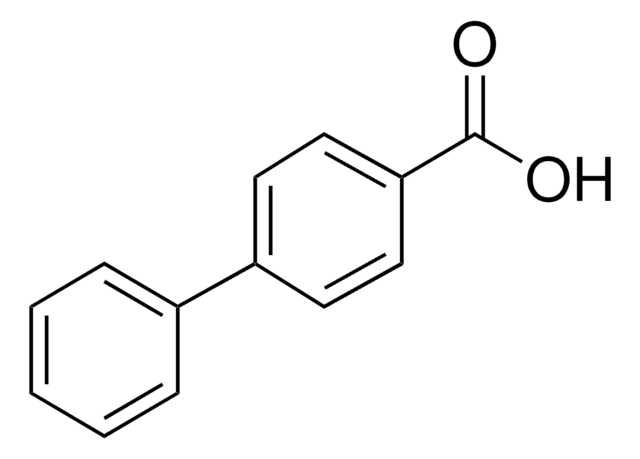


![2-[2-(2-Chloroethoxy)ethoxy]ethanol 96%](/deepweb/assets/sigmaaldrich/product/structures/902/295/ff6d7bb1-a7e0-4582-86e1-819084626e67/640/ff6d7bb1-a7e0-4582-86e1-819084626e67.png)

