All Photos(2)
About This Item
Empirical Formula (Hill Notation):
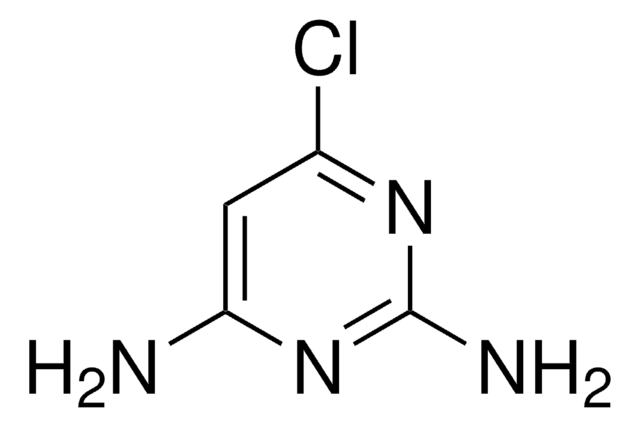
C5H6ClN3
CAS Number:
Molecular Weight:
143.57
Beilstein:
114297
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
mp
183-186 °C (lit.)
solubility
acetic acid: soluble 50 mg/mL, clear, colorless to faintly yellow
functional group
chloro
SMILES string
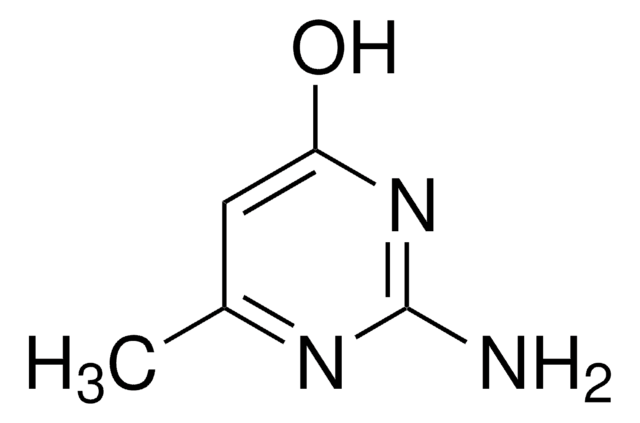
Cc1cc(Cl)nc(N)n1
InChI
1S/C5H6ClN3/c1-3-2-4(6)9-5(7)8-3/h2H,1H3,(H2,7,8,9)
InChI key
NPTGVVKPLWFPPX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Amino-4-chloro-6-methylpyrimidine is a nitification inhibitor.
Application
2-Amino-4-chloro-6-methylpyrimidine was used to study the influence of chlorine substitution in pyrimidine ring on proton donor ability of amino group in 2-aminopyrimidine.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
T Jayavarthanan et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 97, 811-824 (2012-08-21)
The solid phase FTIR and FT-Raman spectra of 2-amino-4-chloro-6-methylpyrimidine (2A4Cl6MP) have been recorded in the regions 400-4000 and 50-4,000 cm(-1), respectively. The spectra have been interpreted interms of fundamentals modes, combination and overtone bands. The structure of the molecule has
J A Hutter et al.
Biochemistry, 26(7), 1969-1973 (1987-04-07)
Thiaminase I from Bacillus thiaminolyticus strain Matsukawa et Misawa is completely and irreversibly inhibited by treatment with 4-amino-6-chloro-2-methylpyrimidine. Inhibition is a time-dependent first-order process, exhibiting a half-time of 4 h at an inhibitor concentration of 5 mM. A specific active-site-directed
Effects of nitrification inhibitors on denitrification of nitrate in soil.
Bremner JM andYeomans JC.
Biology and Fertility of Soils, 2(4), 173-179 (1986)
Christer B Aakeröy et al.
Pharmaceutics, 3(3), 601-614 (2011-01-01)
In the pharmaceutical industry, co-crystals are becoming increasingly valuable as crystalline solids that can offer altered/improved physical properties of an active pharmaceutical ingredient (API) without changing its chemical identity or biological activity. In order to identify new solid forms of
Influence of chlorine-substitution in pyrimidine ring on proton donor ability in H-bond and parameters of amino group of 2-amino pyrimidine.
Borisenko VE, et al.
Vibrational Spectroscopy, 37(1), 97-109 (2005)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service