All Photos(1)
About This Item
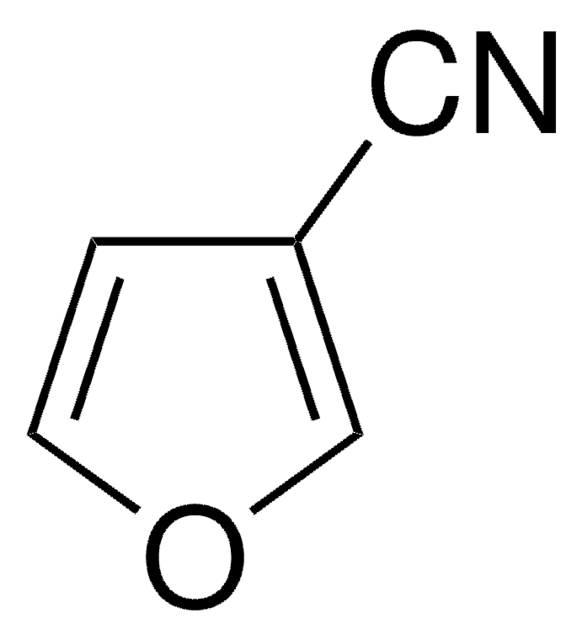
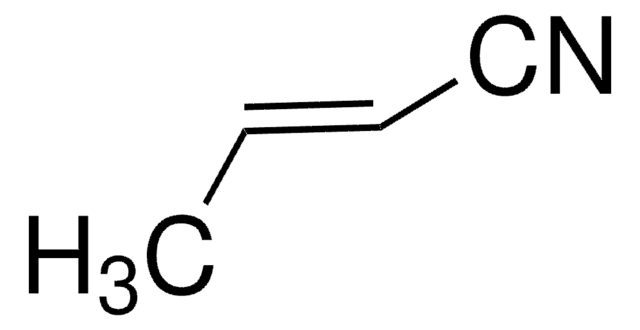
Empirical Formula (Hill Notation):
C5H3NO
CAS Number:
Molecular Weight:
93.08
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
liquid
refractive index
n20/D 1.479 (lit.)
bp
146-148 °C (lit.)
density
1.064 g/mL at 25 °C (lit.)
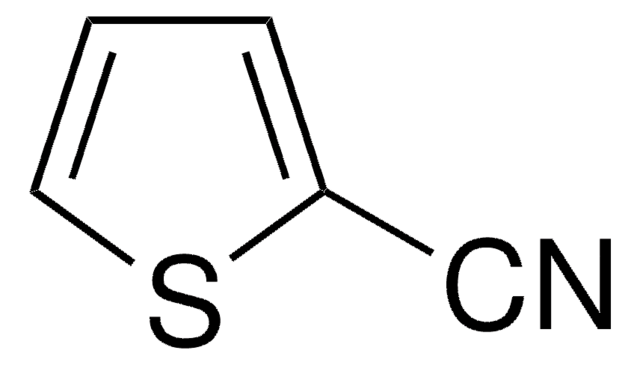
SMILES string
N#Cc1ccco1
InChI
1S/C5H3NO/c6-4-5-2-1-3-7-5/h1-3H
InChI key
YXDXXGXWFJCXEB-UHFFFAOYSA-N
General description
Infrared and Raman spectra of 2-furonitrile has been studied.
Application
2-Furonitrile was employed as substrate to investigate the substrate specificity of nitrilase from Rhodococcus rhodochrous Jl cell.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Dam. 1 - Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
95.0 °F - closed cup
Flash Point(C)
35 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Vibrational spectra of 2-and 3-furonitrile.
Volka K, et al.
Spectrochimica Acta Part A: Molecular Spectroscopy, 32(2), 397-401 (1976)
Nitrilase-Catalyzed Production of Nicotinic Acid from 3-Cyanopyridine in Rhodococcus rhodochrous J1.
C D Mathew et al.
Applied and environmental microbiology, 54(4), 1030-1032 (1988-04-01)
The nitrilase which occurs abundantly in cells of Rhodococcus rhodochrous J1 catalyzes the direct hydrolysis of 3-cyanopyridine to nicotinic acid without forming nicotinamide. By using resting cells, the reaction conditions for nicotinic acid production were optimized. Under the optimum conditions
Masazumi Tamura et al.
Nature communications, 6, 8580-8580 (2015-10-06)
Multidentate materials formed by simply mixing heterogeneous and homogeneous components are promising for construction of versatile active sites on the surface of heterogeneous compounds, however, to the best of our knowledge, there are no reports on such materials. Self-assembly of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service