All Photos(1)
About This Item
Empirical Formula (Hill Notation):
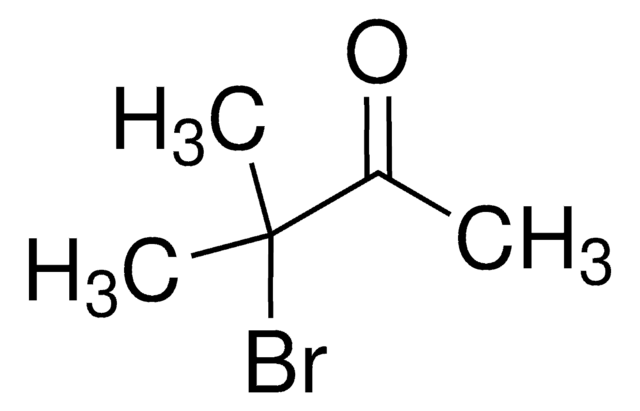
C4H7BrO
CAS Number:
Molecular Weight:
151.00
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
refractive index
n20/D 1.460
density
1.416 g/mL at 25 °C
SMILES string
CC(Br)C(C)=O
InChI
1S/C4H7BrO/c1-3(5)4(2)6/h3H,1-2H3
InChI key
BNBOUFHCTIFWHN-UHFFFAOYSA-N
Application
3-Bromo-2-butanone can be used as a reactant to synthesize:
- N
- -(4,5-Dimethyl-1H-imidazol-2-yl)acetamide by reacting with N-acetylguanidine under acidic conditions.
- Oxazole derivatives by silver triflate catalyzed cyclization reaction with various amides.
- 2-bromo-5,6-dimethyl[1,3]oxazolo[3,2-b][1,2,4]triazole by treating with 3,5-dibromo-1,2,4-triazole in the presence DBU.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Flam. Liq. 3 - Skin Corr. 1B
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
123.8 °F
Flash Point(C)
51 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
[1, 3] Oxazolo [3, 2-b][1, 2, 4] triazoles: a versatile synthesis of a novel heterocycle
Ball Catherine, et al.
Tetrahedron Letters, 51(30), 3907-3909 (2010)
Synthesis of 2, 4-and 2, 4, 5-substituted oxazoles via a silver triflate mediated cyclization
Bailey JL and Sudini RR
Tetrahedron Letters, 55(27) (2014)
Chai Hoon Soh et al.
Journal of combinatorial chemistry, 10(1), 118-122 (2007-12-28)
A microwave-assisted protocol was developed for the construction of di- and monosubstituted 2-aminoimidazoles. The two-step reaction involves the synthesis of N-(1H-imidazol-2-yl)acetamides from readily available alpha-haloketones and N-acetylguanidine, followed by deacetylation. Significant rate enhancement was observed for both steps of the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service