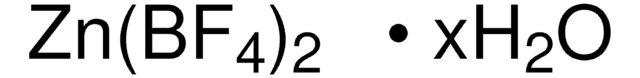705055
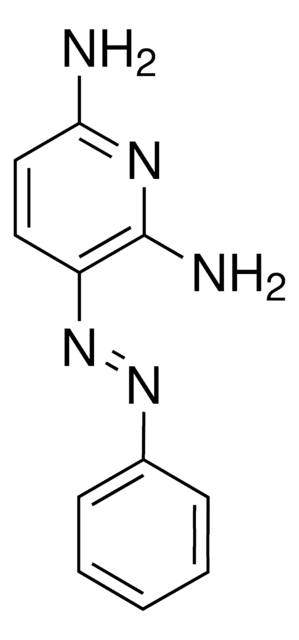
4,4′-Azopyridine
Synonym(s):
Azobis(4-pyridine)
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C10H8N4
CAS Number:
Molecular Weight:
184.20
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
solid
Quality Level
mp
96-101 °C
SMILES string
c1cc(ccn1)N=Nc2ccncc2
InChI
1S/C10H8N4/c1-5-11-6-2-9(1)13-14-10-3-7-12-8-4-10/h1-8H
InChI key
XUPMSLUFFIXCDA-UHFFFAOYSA-N
Related Categories
General description
4,4′-Azopyridine is an aromatic heterocyclic compound used as a ligand in the formation of [HgI2(4,4′-azopyridine)]n complex.
Application
4,4′-Azopyridine can be used:
- To prepare porous coordination polymers (PCPs) by reacting with Zn(NO3)2 and 1,4-benzenedicarboxylic acid.
- As a reagent for the conversion of aliphatic alcohols into disulfides under Mitsunobu conditions.
- To prepare 4,4′-azopyridine-bridged binuclear zinc(II) complexes.
Reactant for preparation of:
Reagent in:
- Flexible porous coordination polymers constructed from 1,2-bis(4-pyridyl)hydrazine via solvothermal in situ reduction reaction
- Metal-organic frameworks based on transition-metal carboxylate clusters as secondary building units
- Heteroaromatic azo compounds under Mitsunobu conditions
- MnII, CoII, and ZnII coordination polymers
- Low-dimensional and porous coordination compounds capable of supramolecular aromatic interactions
Reagent in:
- Facile Mitsunobu esterification reactions
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Heteroaromatic azo compounds as efficient and recyclable reagents for direct conversion of aliphatic alcohols into symmetrical disulfides
Iranpoor N, et al.
Tetrahedron Letters, 53(51), 6913-6915 (2012)
4, 4?-Azopyridine as an easily prepared and recyclable oxidant for synthesis of symmetrical disulfides from thiols or alkyl halides (tosylates)/thiourea}
Khalili, Dariush and Iranpoor
Journal of Sulfur Chemistry, 36, 544-555 (2015)
A novel 4, 4′-azopyridine-bridged binuclear zinc complex
Zhu L, et al.
Journal of Coordination Chemistry, 56(17), 1447-1453 (2003)
A novel HgI2 adduct with an azopyridine ligand: synthesis, structure and optical refractive effect of [HgI2 (4, 4?-azopyridine)] n
Niu, Yunyin and Song
CrystEngComm, 3, 152-154 (2001)
Flexible porous coordination polymers constructed from 1, 2-bis (4-pyridyl) hydrazine via solvothermal in situ reduction of 4, 4′-azopyridine
Liu X, et al.
Dalton Transactions, 40(34), 8549-8554 (2011)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service