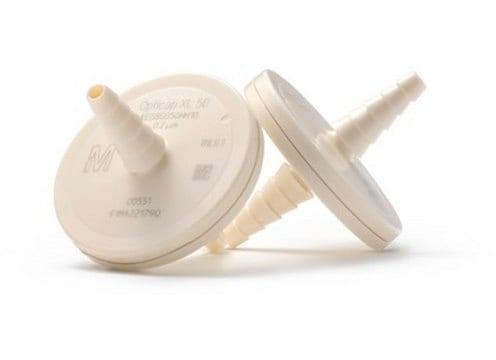
MTGR85010
Aervent® 50 Capsule Filter
pore size 0.2 μm
Synonym(s):
Aervent®-50 Filter Unit 0.2 µm hydrophobic 1/4 in. HB/HB, EPA Est. 041237-FRA-001
About This Item
Recommended Products
material
PTFE membrane
polypropylene
polypropylene housing
Quality Level
sterility
non-sterile
Sterilization Compatibility
gamma compatible
product line
Aervent®
feature
hydrophobic
manufacturer/tradename
Aervent®
parameter
2.1 bar max. differential pressure at 25 °C
4.1 bar max. inlet pressure (60 psi) at 25 °C
technique(s)
gas filtration: suitable
L
7.2 cm (2.84 in.)
W
2.4 in.
diam.
6.2 cm (2.4 in.)
filtration area
19.6 cm2
impurities
≤0.5 EU/mL (LAL test, Aqueous extraction)
gravimetric extractables
≤1.0 mg
matrix
Aervent®
pore size
0.2 μm pore size
bubble point
≥1100 mbar (16 psig), nitrogen with 60/40% IPA/water at 23 °C
fitting
inlet/outlet hose for 6.5-9.5 mm I.D. silicone tubing
shipped in
ambient
General description
Application
- Sterile venting of vessels and carboys
- Autoclave vacuum break lines
- Low volume sterile filtration of non-aqueous fluids
- Small fermenter inlet and exhaust gas filtration
Preparation Note
10 autoclave cycles of 30 min @ 130 °C; not in-line steam sterilizable
Analysis Note
Quantitative retention of 107 CFU/cm2Brevundimonas diminuta (ATCC® 19146) per ASTM F838-83 methodology
Other Notes
- Organism Retention: Microorganism
- Mode of Action: Filtration (size exclusion)
- Application: BioProcessing
- Intended Use: Reduction or removal of microorganism/bioburden
- Instructions for Use: Please refer Steam Sterilization & Integrity Testing Procedures guide and first page of Visual Inspection Guide
- Storage Statement: Store in dry location
- Disposal Statement: Dispose of in accordance with applicable federal, state and local regulations.
Legal Information
Not finding the right product?
Try our Product Selector Tool.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service