L6150
Lysine Oxidase from Trichoderma viride
lyophilized powder, ≥20 units/mg protein
Synonym(s):
L-Lysine:oxygen oxidoreductase (deaminating)
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
CAS Number:
MDL number:
UNSPSC Code:
12352204
NACRES:
NA.54
Recommended Products
biological source
fungus (Trichoderma viride)
Quality Level
form
lyophilized powder
specific activity
≥20 units/mg protein
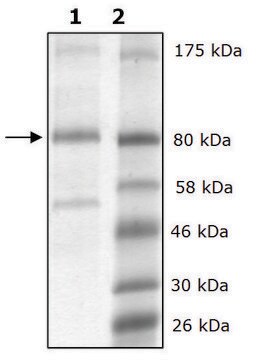
mol wt
112 kDa
composition
Protein, 5-20%
storage temp.
2-8°C
General description
Lysine Oxidase from Trichoderma viride is a homodimeric flavoenzyme corresponding to molecular mass of 112 kDa. It is stable at 65°C and is highly specific for L-lysine substrate. It comprises FAD-binding, substrate binding and a helical domain with distinct active site funnel.
Application
Lysine Oxidase from Trichoderma viride has been used in the preparation of luminescent biochip preparation.
Biochem/physiol Actions
Lysine Oxidase from Trichoderma viride catalyzes the formation of α-keto- ε-aminocaproate by the oxidative deamination of L-lysine. It displays anti-tumor functionality in cancer leukaemic cells. It is a tumor suppressor for squamous cell, fibroblast, ovarian and gastric tumors. Lysine oxidase also plays key role in connective tissue structural integrity and embryo development.
Unit Definition
One unit will catalyze the formation of 1 μmole of 6-amino-2-oxohexanoic acid from L-lysine per min at 37°C at pH 8.0.
Physical form
Contains phosphate buffer salts and stabilizer
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Raluca-Ioana Stefan-van Staden et al.
Biosensors & bioelectronics, 35(1), 439-442 (2012-03-16)
An amperometric biosensor was proposed for the enantioanalysis of L-lysine. The biosensor is based on the impregnation of L-lysine oxidase in diamond paste. The potential used for the determination of l-lysine was 650 mV. The biosensor exhibited a linear concentration
Studies on Anti-Cancer Activity of Lysyl Oxidase from Trichoderma Viride MTCC 167
Kalra S, et al.
International Journal of Applied Sciences and Biotechnology, 4(1), 57-63 (2016)
Reza Karimi Shervedani et al.
Bioelectrochemistry (Amsterdam, Netherlands), 75(2), 124-129 (2009-04-07)
Immobilization of L-lysine alpha-oxidase on gold-mercaptopropionic acid self-assembled monolayer (Au-MPA-LOx SAM) electrode is verified experimentally in the present work. Fabrication steps and electrochemical interaction of Au-MPA-LOx with L-lysine were monitored by general electrochemical methods like cyclic voltammetry (CV) and chronoamperometry
I P Smirnova et al.
Voprosy meditsinskoi khimii, 44(4), 384-387 (1998-12-10)
L-lysin-a-oxidase (LO), a fungal enzyme catalysing oxidative deamination of L-lysin, was used for the inhibition of the reproduction of herpes simplex virus type 1 (HSV-1). Antiviral activity of LO was tested in vitro. The expression of viral antigens and CPE
Design of luminescent biochips based on enzyme, antibody, or DNA composite layers
Marquette CA, et al.
Analytical and Bioanalytical Chemistry, 377(5), 922-928 (2003)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service