377953
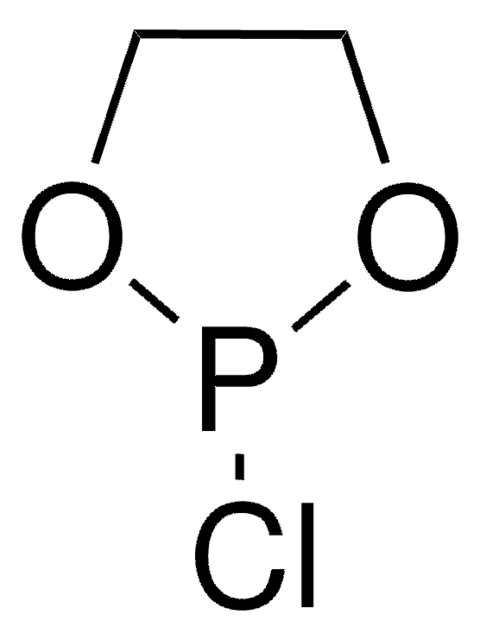
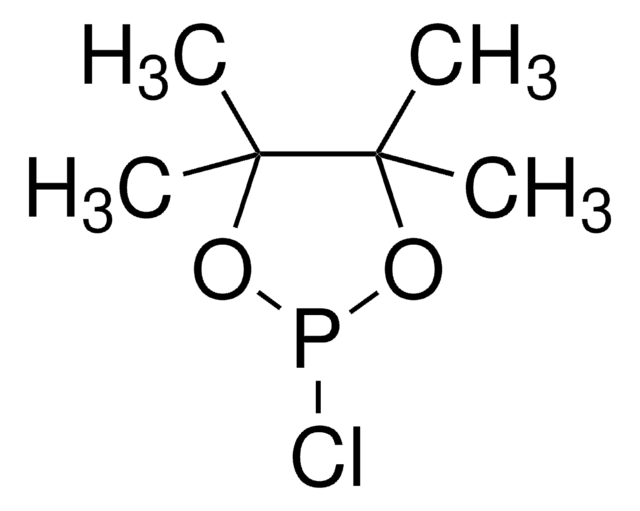
2-Chloro-1,3,2-dioxaphospholane 2-oxide
Synonym(s):
2-Chloro-2-oxo-1,3,2-dioxaphospholane, Ethylene glycol chlorophosphate
About This Item
Recommended Products
form
liquid
impurities
<10% 2-Chloro-1,3,2-dioxaphospholane
refractive index
n20/D 1.45 (lit.)
bp
89-91 °C/0.8 mmHg (lit.)
mp
12-14 °C (neat) (lit.)
density
1.55 g/mL at 25 °C (lit.)
storage temp.
−20°C
SMILES string
ClP1(=O)OCCO1
InChI
1S/C2H4ClO3P/c3-7(4)5-1-2-6-7/h1-2H2
InChI key
SBMUNILHNJLMBF-UHFFFAOYSA-N
Related Categories
General description
2-Chloro-1,3,2-dioxaphospholane 2-oxide is used in esterification reactions for cyclic phosphate synthesis, also reacts with phenyl grignard reagents.
Application
- 2-methacryloyloxyethylphosphorylcholine
- miltefosine (hexadecylphosphocholine, MT) analogs
- phosphoric acid 2-trimethylamino-ethyl ester undec-10-enyl ester
- uridine nucleolipid, (2′,3′-O-16-hentriacontanyliden-uridine-5′-phosphocholine, PUPC)
- adenosine nucleoamphiphile, (2′,3′-O-16-hentriacontanyliden-adenosine-5′-phosphocholine, PAPC)
- structurally related phospholipids which are either conformationally restricted or flexible
- phosphatidylcholines
- Synthesis of amino-functionalized hybrid hydrocarbon/fluorocarbon double-chain phospholipid
- Synthesis of UV-polymerizable lipids via Chabrier reaction
- Syntheses of block copolymers of poly(aliphatic ester) with clickable polyphosphoester
- Imprinting molecular recognition sites on multiwalled carbon nanotubes surface for electrochemical detection of insulin in real samples
- Synthesis of a zwitterionic silane
- Synthesis of a core-shell-corona micelle stabilized by reversible cross-linkage for intracellular drug delivery
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Supplementary Hazards
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
>235.4 °F - closed cup
Flash Point(C)
> 113 °C - closed cup
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service