M-020
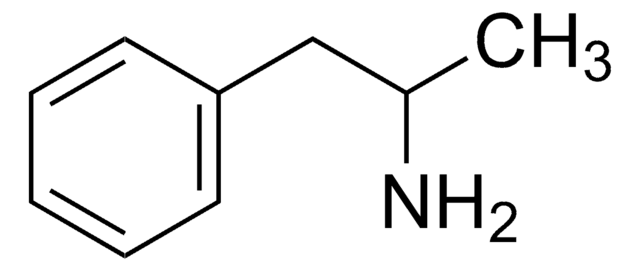
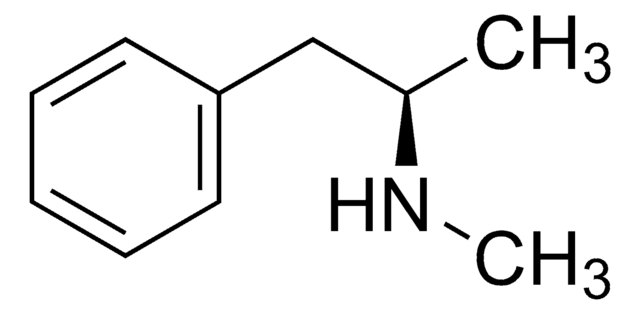
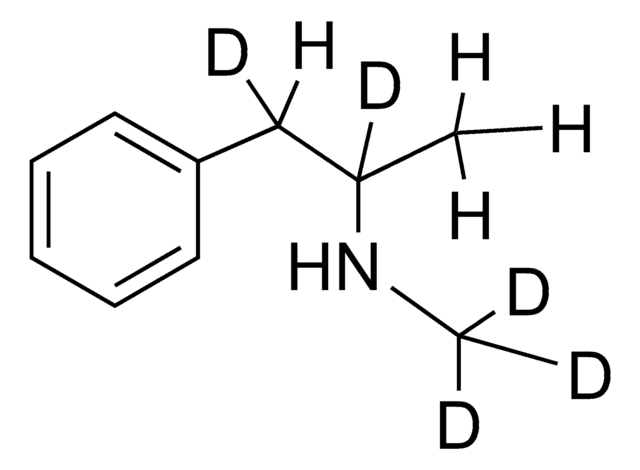
S(+)-Methamphetamine solution
1.0 mg/mL in methanol, ampule of 1 mL, certified reference material, Cerilliant®
Synonym(s):
dextro-Methamphetamine
About This Item
Recommended Products
grade
certified reference material
Quality Level
form
liquid
feature
Snap-N-Spike®/Snap-N-Shoot®
packaging
ampule of 1 mL
manufacturer/tradename
Cerilliant®
drug control
Narcotic Licence Schedule A (Switzerland); psicótropo (Spain); Decreto Lei 15/93: Tabela IIB (Portugal)
concentration
1.0 mg/mL in methanol
technique(s)
gas chromatography (GC): suitable
liquid chromatography (LC): suitable
application(s)
forensics and toxicology
format
single component solution
storage temp.
2-8°C
SMILES string
CN[C@@H](C)Cc1ccccc1
InChI
1S/C10H15N/c1-9(11-2)8-10-6-4-3-5-7-10/h3-7,9,11H,8H2,1-2H3/t9-/m0/s1
InChI key
MYWUZJCMWCOHBA-VIFPVBQESA-N
Gene Information
human ... SLC6A2(6530) , SLC6A3(6531) , SLC6A4(6532)
General description
Application
- Pharmacokinetic Studies of S(+)-Methamphetamine: A comprehensive study assessing the pharmacokinetic profile of S(+)-Methamphetamine highlighted its rapid absorption and metabolism, offering critical insights into its therapeutic potential and challenges in drug abuse scenarios. This research aids in optimizing dosing regimens and understanding the drug′s behavior in biological systems, essential for both clinical applications and forensic investigations (Rushton et al., 2024).
- Pharmaceutical Applications of S(+)-Methamphetamine: Research on S(+)-Methamphetamine′s role in neurochemical processes explored its effects on neurotransmitter systems, particularly focusing on its potential therapeutic applications for attention deficit hyperactivity disorder (ADHD) and weight loss. Studies like these inform drug design and repurposing strategies, helping to mitigate abuse risks while harnessing pharmacological benefits (Pardo et al., 2024).
- Drug Metabolism and Enantiomeric Analysis: Investigations into the metabolism of S(+)-Methamphetamine have provided detailed insights into its biotransformation pathways, crucial for developing antidotes and treatment strategies for overdose cases. This research also enhances the understanding of enantiomeric differences in drug activity, supporting the development of more targeted and effective therapeutic agents (Liut et al., 2024).
- Clinical Trials and Neurological Research: Systematic reviews of clinical trials involving S(+)-Methamphetamine have elucidated its complex pharmacodynamics and associated behavioral effects. This information is vital for neuroscientists and pharmacologists working to develop safer and more effective CNS stimulants with fewer side effects and lower abuse potential (Barkholtz et al., 2023).
Legal Information
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Flam. Liq. 2 - STOT SE 1
Target Organs
Eyes
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point(F)
49.5 °F - closed cup
Flash Point(C)
9.7 °C - closed cup
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Optimized sample prep and chiral chromatography methods for the LC/MS analysis of these drug enantiomers in urine
In this study, optimized methods are presented for sample preparation and chiral chromatography for the LC/MS analysis of amphetamine and methamphetamine enantiomers in urine.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service