11080725001
Roche
Neuraminidase (Sialidase)
from Vibrio cholerae
Synonym(s):
Salidase
About This Item
Recommended Products
biological source
Vibrio cholerae
Quality Level
form
solution
mol wt
~95 kDa
packaging
pkg of 1 U
manufacturer/tradename
Roche
optimum pH
5.5-6.2
suitability
suitable for ELISA applications
application(s)
life science and biopharma
sample preparation
shipped in
wet ice
storage temp.
2-8°C
General description
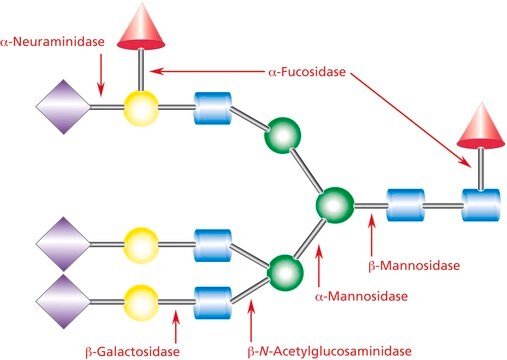
Specificity
Application
- to remove cis-acting sialic acids in CHO (chinese hamster ovary) cells
- for deglycosylation studies
Unit Definition
10 mM N-acetyl-neuraminosyl-D-lactose, 50 mM sodium acetate, 4 mM calcium chloride, bovine serum albumin, 100 μg/ml, pH 5.5. The activity is determined by measuring the released D-lactose using the β-galactosidase/galactose dehydrogenase method. Under the same conditions, 1 μmol N-acetylneuraminic acid per min is split off from human acid α1-glycoprotein (10 mg/ml incubation mixture) by 1 U neuraminidase. Released N-acetyl-neuraminic acid can be determined using, for example, the thiobarbituric acid method.
Physical form
Note: The serum used for this preparation was tested for HBs antigen and for the presence of antibodies to HIV-1, HIV-2, HCV, and found to be negative, according to the current quality control procedures.
Other Notes
Legal Information
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Sens. 1
Storage Class Code
12 - Non Combustible Liquids
WGK
WGK 1
Flash Point(F)
does not flash
Flash Point(C)
does not flash
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Neuraminidase can be used to cleave sialic acids from proteins. In this protocol, the enzyme from Vibrio cholerae is used on fixed cells.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service