A2411
Amphotericin B from Streptomyces sp.
solid, suitable for cell culture, BioReagent
Synonym(s):
Fungizone
About This Item
Recommended Products
product name
Amphotericin B from Streptomyces sp., BioReagent, suitable for cell culture, ~80% (HPLC)
biological source
Streptomyces sp.
Quality Level
product line
BioReagent
Assay
~80% (HPLC)
form
solid
potency
>750.00 μg per mg (Dry basis)
technique(s)
cell culture | mammalian: suitable
color
yellow to yellow-orange
solubility
H2O: slightly soluble
antibiotic activity spectrum
fungi
yeast
Mode of action
cell membrane | interferes
storage temp.
2-8°C
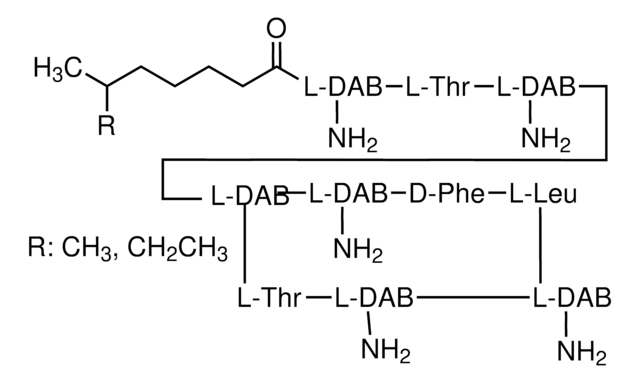
SMILES string
C[C@H]1O[C@@H](O[C@@H]2C[C@@H]3O[C@](O)(C[C@@H](O)C[C@@H](O)[C@H](O)CC[C@@H](O)C[C@@H](O)CC(=O)O[C@@H](C)[C@H](C)[C@H](O)[C@@H](C)\C=C\C=C\C=C\C=C\C=C\C=C\C=C\2)C[C@H](O)[C@H]3C(O)=O)[C@@H](O)[C@@H](N)[C@@H]1O
InChI
1S/C47H73NO17/c1-27-17-15-13-11-9-7-5-6-8-10-12-14-16-18-34(64-46-44(58)41(48)43(57)30(4)63-46)24-38-40(45(59)60)37(54)26-47(61,65-38)25-33(51)22-36(53)35(52)20-19-31(49)21-32(50)23-39(55)62-29(3)28(2)42(27)56/h5-18,27-38,40-44,46,49-54,56-58,61H,19-26,48H2,1-4H3,(H,59,60)/b6-5+,9-7+,10-8+,13-11+,14-12+,17-15+,18-16+/t27-,28-,29-,30+,31+,32+,33-,34-,35+,36+,37-,38-,40+,41-,42+,43+,44-,46-,47+/m0/s1
InChI key
APKFDSVGJQXUKY-INPOYWNPSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
Biochem/physiol Actions
Caution
Preparation Note
Other Notes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
STOT RE 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Prevent fungal, yeast, and mold contamination in cell cultures. Discover the best antifungal agent for your cultures with the extensive Sigma® antifungal collection.
Antibiotic kill curve is a dose response experiment in which mammalian cells are subjected to increasing amounts of selection antibiotic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service