UC179
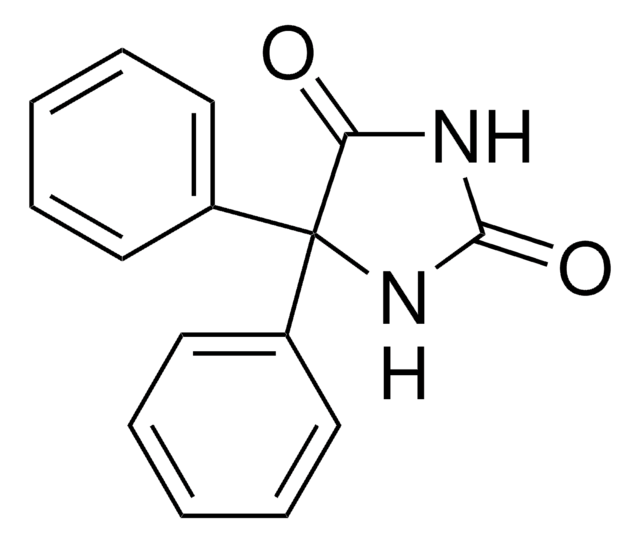
(R)-(−)-Nirvanol
Synonym(s):
(R)-(−)-5-Ethyl-5-phenyl-2,4-imidazolidinedione, (R)-(−)-5-Ethyl-5-phenylhydantoin
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C11H12N2O2
CAS Number:
Molecular Weight:
204.23
MDL number:
UNSPSC Code:
12161501
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
form
solid
Quality Level
color
off-white
solubility
DMF: soluble
storage temp.
2-8°C
SMILES string
CC[C@@]1(NC(=O)NC1=O)c2ccccc2
InChI
1S/C11H12N2O2/c1-2-11(8-6-4-3-5-7-8)9(14)12-10(15)13-11/h3-7H,2H2,1H3,(H2,12,13,14,15)/t11-/m1/s1
InChI key
UDTWZFJEMMUFLC-LLVKDONJSA-N
Biochem/physiol Actions
CYP2B6 metabolite of (S)-(+)-mephenytoin; anticonvulsant; hypnotic.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
Preparation Note
Nirvanol is soluble in DMF.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
A Pezeshk et al.
Journal of inorganic biochemistry, 42(4), 267-272 (1991-06-01)
The preparation and spectral properties of copper(II) complexes of two hydantoins are reported. Complexes of the general formula Cu(hyd)2(py)2, where hyd = phenytoin or nirvanol; and py = pyridine were prepared and characterized by infrared and ESR. Spectral data show
B F Bourgeois et al.
Epilepsia, 27(4), 412-418 (1986-07-01)
Stereoselective metabolism has been demonstrated for mephenytoin (MHT), R-MHT being demethylated to the pharmacologically active metabolite 5-phenyl-5-ethylhydantoin (PEH; nirvanol), and S-MHT undergoing aromatic hydroxylation to 4-OH-MHT, with formation of an intermediate arene oxide metabolite. PEH is responsible for the therapeutic
H Fujioka et al.
Journal of pharmacobio-dynamics, 9(3), 303-314 (1986-03-01)
Disposition of 1-benzenesulfonyl-5,5-diphenylhydantoin (II) having a potent anti-inflammatory activity was compared with that of 5,5-diphenylhydantoin (I), an antiepileptic drug, in order to elucidate whether the pharmacodynamic difference between them can be explained by their physicochemical and pharmacokinetic properties. After oral
R W Nims et al.
Journal of biochemical toxicology, 9(6), 279-288 (1994-12-01)
To explore the enantioselectivity of ligand interaction with the putative phenobarbital receptor, the pharmacodynamics of cytochrome P450 2B (CYP2B) induction by racemic 5-ethyl-5-phenylhydantoin and its two enantiomers were investigated in the male F344/NCr rat and in cultured adult male rat
R W Nims et al.
Chemical research in toxicology, 6(2), 188-196 (1993-03-01)
The pharmacodynamics of rat hepatic cytochrome P450 2B (P450 2B) induction by phenobarbital (PB) and two structural congeners, dl-5-ethyl-5-phenylhydantoin (EPH) and dl-5-ethyl-5-phenyloxazolidinedione (EPO), were investigated. The in vivo induction of P450 2B was probed in F344/NCr rats by measuring immunoreactive
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service