125504
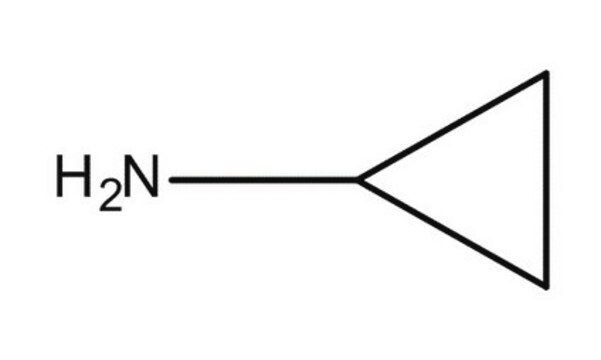
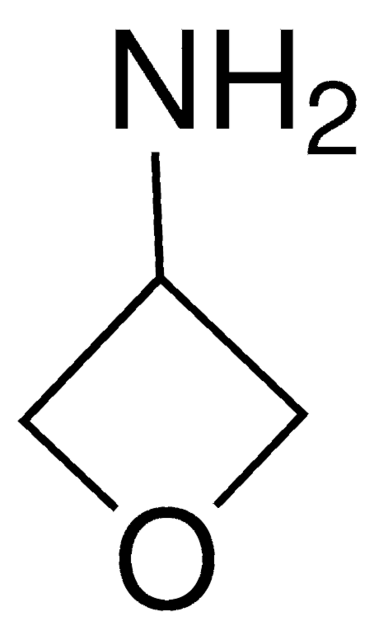
Cyclopropylamine
98%
Synonym(s):
Aminocyclopropane
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C3H5NH2
CAS Number:
Molecular Weight:
57.09
Beilstein:
741858
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
vapor pressure
4.67 psi ( 20 °C)
Quality Level
Assay
98%
form
liquid
autoignition temp.
527 °F
refractive index
n20/D 1.420 (lit.)
bp
49-50 °C (lit.)
density
0.824 g/mL at 25 °C (lit.)
SMILES string
NC1CC1
InChI
1S/C3H7N/c4-3-1-2-3/h3H,1-2,4H2
InChI key
HTJDQJBWANPRPF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Cyclopropylamine(CPA) has been used in the synthesis of N-[4-(4-fluoro)phenyl-2-aminothiazol-5-yl]pyrimidin-2-yl-alkylamine derivatives. It has been used in the synthesis of Pt(CPA)2(bismethylthiomethylenepropanedioate) and Pt(CPA)2(bisethylthiomethylenepropanedioate) complexes.
Biochem/physiol Actions
Cyclopropylamine inactivates cytochrome P450 enzymes by a mechanism involving initial one-electron oxidation at nitrogen followed by scission of the cyclopropane ring leading to covalent modification of the enzyme. It is a mechanism-based inhibitor of quinoprotein methylamine dehydrogenase from Paracoccus denitrificans.
accessory
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Flam. Liq. 2 - Skin Corr. 1B
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
33.8 °F - closed cup
Flash Point(C)
1 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Coordination Mode vs. Anticancer Activity of the Platinum (II) Complexes Involving Sulfur-Containing Ylidenemalonate Ligands.
Sakai N, et al.
Bull. Korean Chem. Soc., 19(12), 1377-1379 (1998)
Intermolecular [3+2] cycloaddition of cyclopropylamines with olefins by visible-light photocatalysis.
Soumitra Maity et al.
Angewandte Chemie (International ed. in English), 51(1), 222-226 (2011-11-24)
Ana Paula B Vintém et al.
Bioorganic & medicinal chemistry, 13(10), 3487-3495 (2005-04-26)
Modification of cysteine (Cys) residues inactivates monoamine oxidases (MAO) yet the crystal structure shows no conserved cysteines in the active site of MAO A (Ma, J. et al. J. Mol. Biol.2004, 338, 103-114). MAO A cysteine 374 was mutated to
Qin Sun et al.
Chemical research in toxicology, 21(3), 711-719 (2008-02-27)
Trovafloxacin (Trovan) is a fluoroquinolone antibiotic drug with a long half-life and broad-spectrum activity. Since its entry into the market in 1998, trovafloxacin has been associated with numerous cases of hepatotoxicity, which has limited its clinical usefulness. Trovafloxacin possesses two
Etienne Veignie et al.
Environmental pollution (Barking, Essex : 1987), 129(1), 1-4 (2004-01-30)
In order to study the enzymatic mechanisms involved in the successive steps of BaP degradation by a Deuteromycete fungus Fusarium solani, we developed an indirect approach by using inhibitors of enzymes. We used either specific inhibitors of peroxidases (i.e. salicylhydroxamic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service