G4155
β-Galactosidase from Escherichia coli
aqueous glycerol suspension, ≥500 units/mg protein (biuret)
Synonym(s):
β-D-Galactoside galactohydrolase, Lactase
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
CAS Number:
MDL number:
UNSPSC Code:
12352204
NACRES:
NA.54
Recommended Products
form
aqueous glycerol suspension
Quality Level
specific activity
≥500 units/mg protein (biuret)
mol wt
465 kDa
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
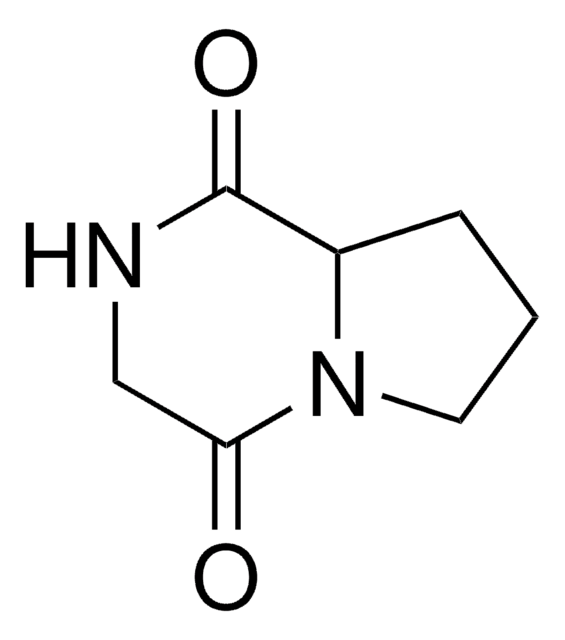
β-Galactosidase is used in the enzymatic assays in the synthesis of imidazolo-pyrrolidinoses.
Biochem/physiol Actions
β-galactosidase cleaves lactose into its monosaccharide components, glucose and galactose. It also catalyses the transglycosylation of glucose into allolactose, the inducer of β-galactosidase, in a feedback loop.
Physical properties
Tetramer molecular weight 465 kDa (subunits 116.3 kDa each)
Unit Definition
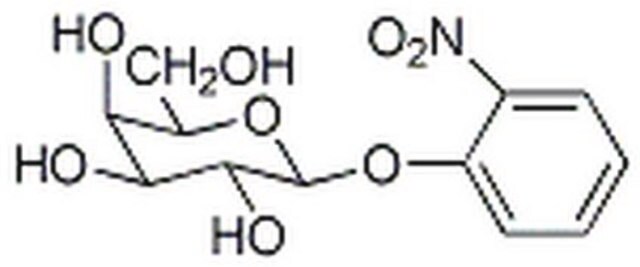
One unit will hydrolyze 1.0 μmole of o-nitrophenyl β-D-galactoside to o-nitrophenol and D-galactose per min at pH 7.3 at 37 °C.
Physical form
Suspension in 50% glycerol, 5 mM Tris buffer salts, 5 mM magnesium chloride, 0.5 mM DTT, 0.5 mM mercaptoethanol. pH 7.4
Other Notes
Loss of enzymatic activity can occur if this enzyme is frozen. This enzyme is provided in a 50% glycerol solution and will not freeze if stored at -20° Celsius. It is not recommended that this enzyme preparation be stored in an ultracold freezer (-60 to -100° Celsius).
inhibitor
Product No.
Description
Pricing
substrate
Product No.
Description
Pricing
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Théophile Tschamber et al.
Bioorganic & medicinal chemistry, 11(17), 3559-3568 (2003-08-07)
The syntheses of four glyco-imidazoles, which are pentose-derivatives belonging to the D-series, as well as the syntheses of their L-enantiomers, are reported. Starting from the known linear xylo, lyxo, arabino, and ribo imidazolo-pentoses in both the L- and the D-series
Olga Ermakova et al.
Brain sciences, 11(6) (2021-07-03)
Acquisition of detailed anatomical and molecular knowledge from intact biological samples while preserving their native three-dimensional structure is still a challenging issue for imaging studies aiming to unravel a system's functions. Three-dimensional micro-CT X-ray imaging with a high spatial resolution
J W Callahan
Biochimica et biophysica acta, 1455(2-3), 85-103 (1999-11-26)
GM1 gangliosidosis and Morquio B disease are distinct disorders both clinically and biochemically yet they arise from the same beta-galactosidase enzyme deficiency. On the other hand, galactosialidosis and sialidosis share common clinical and biochemical features, yet they arise from two
A Hinek et al.
The Journal of clinical investigation, 91(3), 1198-1205 (1993-03-01)
We and others have previously shown that a 67-kD cell surface elastin/laminin-binding protein (EBP) is responsible for cell adhesion to elastin and laminin and for mediating the process of elastin fiber assembly, but the nature of this protein was unknown.
S Privitera et al.
The Journal of biological chemistry, 273(11), 6319-6326 (1998-04-16)
Our previous studies showed immunological and functional similarities, as well as partial sequence homology, between the enzymatically inactive alternatively spliced variant of human beta-galactosidase (S-gal) and the 67-kDa elastin/laminin-binding protein (EBP) from sheep. To define the genetic origin of the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service