All Photos(1)
About This Item
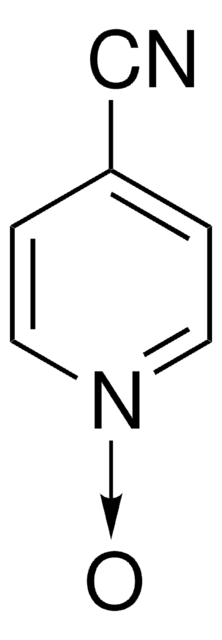
Empirical Formula (Hill Notation):
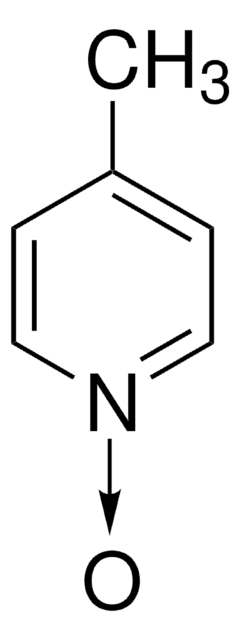
C5H5NO
CAS Number:
Molecular Weight:
95.10
Beilstein:
105257
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
form
solid
bp
270 °C (lit.)
mp
62-67 °C (lit.)
SMILES string
[O-][n+]1ccccc1
InChI
1S/C5H5NO/c7-6-4-2-1-3-5-6/h1-5H
InChI key
ILVXOBCQQYKLDS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Pyridine N-oxide axle with [2]rotaxanes was synthesized via an anion templated threading-followed-by-stoppering strategy.
Application
Pyridine N-oxide was used to study the FTIR spectra of pyridine N-oxide in acetonitrile.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
289.4 °F - closed cup
Flash Point(C)
143 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Takahiko Kojima et al.
Journal of the American Chemical Society, 133(44), 17901-17911 (2011-09-29)
Ruthenium(II)-acetonitrile complexes having η(3)-tris(2-pyridylmethyl)amine (TPA) with an uncoordinated pyridine ring and diimine such as 2,2'-bipyridine (bpy) and 2,2'-bipyrimidine (bpm), [Ru(II)(η(3)-TPA)(diimine)(CH(3)CN)](2+), reacted with m-chloroperbenzoic acid to afford corresponding Ru(II)-acetonitrile complexes having an uncoordinated pyridine-N-oxide arm, [Ru(II)(η(3)-TPA-O)(diimine)(CH(3)CN)](2+), with retention of the coordination
Yichen Tan et al.
Journal of the American Chemical Society, 134(8), 3683-3686 (2012-02-09)
Direct arylations of pyridine N-oxide (PyO), a convenient method to prepare 2-arylpyridines, catalyzed by Pd(OAc)(2) and PtBu(3) have been proposed to occur by the generation of a PtBu(3)-ligated arylpalladium acetate complex, (PtBu(3))Pd(Ar)(OAc) (1), and the reaction of this complex with
Allyn T Londregan et al.
Organic letters, 13(7), 1840-1843 (2011-03-08)
A general and facile one-pot procedure for the synthesis of 2-substituted pyridines from the corresponding pyridine-N-oxides and nucleophiles is presented as a mild alternative to S(N)Ar chemistry. A variety of nucleophiles and heterocyclic-N-oxides participate effectively in this transformation, which uses
Wei Liu et al.
Organic letters, 15(18), 4682-4685 (2013-09-12)
A highly efficient protocol for C-H/C-H cross-coupling has been found to occur between 2-aryl-1,2,3-triazole N-oxides and pyridine N-oxide derivatives. In addition, two homocoupling reactions of 2-substituted 1,2,3-triazole N-oxides and some pyridine N-oxide derivatives were developed. A possible pathway of C-H/C-H
Jan Balzarini et al.
The Journal of antimicrobial chemotherapy, 55(2), 135-138 (2005-01-15)
Pyridine N-oxide derivatives represent a new class of anti-HIV compounds, for which some members exclusively act through inhibition of HIV-1 reverse transcriptase and thus characteristically behave as non-nucleoside reverse transcriptase inhibitors. Other members act, additionally or alternatively, at a post-integrational
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service