All Photos(2)
About This Item
Empirical Formula (Hill Notation):
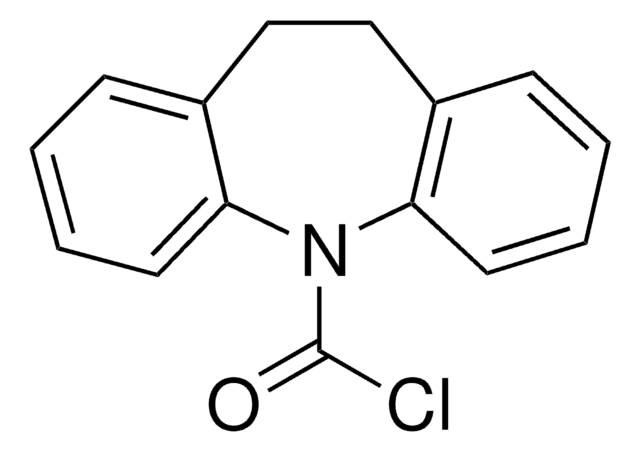
C15H10ClNO
CAS Number:
Molecular Weight:
255.70
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
90%
mp
149-153 °C (lit.)
SMILES string
ClC(=O)N1c2ccccc2C=Cc3ccccc13
InChI
1S/C15H10ClNO/c16-15(18)17-13-7-3-1-5-11(13)9-10-12-6-2-4-8-14(12)17/h1-10H
InChI key
APJYHXJGXDPGBA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Dibenz [b,f]azepine-5-carbonyl chloride or 5H-dibenz [b,f]azepine-5-carbonyl chloride is a tricyclic heterocyclic compound that can be synthesized from 5H-dibenz[ b,f]azepine.
Application
Dibenz [b,f]azepine-5-carbonyl chloride may be used in the preparation of trans-10,11-dibromo-10,11-dihydro-5H-dibenz[b,f]azepine-5-carbonyl chloride via bromination using bromine. It may also be used to prepare urea derivatives, which are potent P2X4 receptor (purinergic receptor) antagonists.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Kinetic evidence for rate determination during the nucleophilic step of olefin bromination. The case of 5H-dibenz [b, f] azepine-5-carbonyl chloride.
Bellucci G and Chiappe C.
The Journal of Organic Chemistry, 58(25), 7120-7127 (1993)
Maoqun Tian et al.
Bioorganic & medicinal chemistry, 22(3), 1077-1088 (2014-01-15)
Antagonists for the P2 receptor subtype P2X4, an ATP-activated cation channel receptor, have potential as novel drugs for the treatment of neuropathic pain and other inflammatory diseases. In the present study, a series of 47 carbamazepine derivatives including 32 novel
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![10,11-Dihydro-5H-dibenz[b,f]azepine 97%](/deepweb/assets/sigmaaldrich/product/structures/282/468/27ed6f23-3d01-4628-8293-f0051a6f3b7c/640/27ed6f23-3d01-4628-8293-f0051a6f3b7c.png)