C6165
Carbonic Anhydrase Isozyme II human
≥80%, powder, ≥3,000 W-A units/mg protein
Synonym(s):
Carbonate Dehydratase, Carbonate Hydrolyase
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
Related Categories
Application
Human carbonic anhydrase isozyme II has been used to assess its gene fusion abilities for efficient expression and recovery of recombinant proteins. Human carbonic anhydrase isozyme II has also been used to investigate a new process for the synthesis of difluoromethanesulfonamides. Furthermore, CA II from Sigma has been used as a standard to measure the CA activity in lung tissue homogenates. The study analysed the possible relationship between the expression of carbonic anhydrase and non-small cell lung cancer. The product has also been used with Freund′s complete adjuvant at 1:1 ratio for antibody production. This study evaluated the quantitative and functional alterations of cytosolic CA isoenzymes in the erythrocytes of glucose-6-phosphate dehydrogenase (G6PD)-deficient individuals. The enzyme has also been used in the study of natural phenolic inhibitors of CA II.
Biochem/physiol Actions
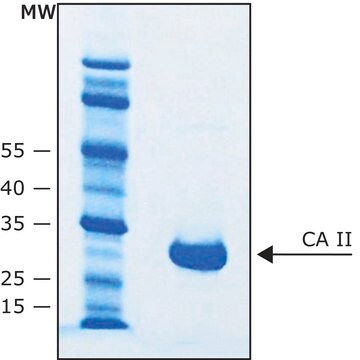
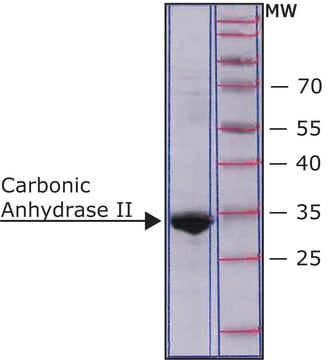
Carbonic anhydrase is a zinc metalloenzyme that catalyzes the hydration of carbon dioxide to carbonic acid. It is involved in pathological and physiological processes such as pH and CO2 homeostasis, transport of bicarbonate and CO2, biosynthetic reactions, bone resorption, calcification, and tumorigenicity. Therefore, this enzyme is an important target for inhibitors that can be used for treating various pathological conditions such as glaucoma, epilepsy and Parkinson′s disease. CAII has a molecular mass of approximately 30 kDa and is primarily present in type II pneumocytes. Due to this unique location it has been speculated that CAII is involved in pulmonary functions such as regulation of fluid secretion and CO2 elimation. Sulfonamides, sulfamates and sulfamides are potent inhibitors of CA.
Unit Definition
One Wilbur-Anderson (W-A) unit will cause the pH of a 0.02 M Trizma buffer to drop from 8.3 to 6.3 per min at 0 °C. (One W-A unit is essentially equivalent to one Roughton-Booth unit.)
inhibitor
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Shinya Tsukiji et al.
Methods in molecular biology (Clifton, N.J.), 1266, 243-263 (2015-01-07)
Introducing nongenetically encoded, synthetic probes into specific proteins is now recognized as a key component in chemical biology. In particular, the ability to chemically modify specific "native" proteins in various contexts from in vitro to cellular systems is of fundamental
Ghita Ghislat et al.
Biomolecules, 10(11) (2020-11-25)
Background and purpose: Identifying the macromolecular targets of drug molecules is a fundamental aspect of drug discovery and pharmacology. Several drugs remain without known targets (orphan) despite large-scale in silico and in vitro target prediction efforts. Ligand-centric chemical-similarity-based methods for
W L Chiang et al.
Clinica chimica acta; international journal of clinical chemistry, 314(1-2), 195-201 (2001-11-24)
This study was designed to evaluate the quantitative and activity alterations of cytosolic carbonic anhydrase (CA) isoenzymes in the erythrocytes of glucose-6-phosphate dehydrogenase (G6PD)-deficient individuals. Western Blot and CA esterase activity analysis were employed to measure cytosolic erythrocyte CA isoenzymes.
Screening and docking studies of natural phenolic inhibitors of carbonic anhydrase II
Huang HQ, et al.
Science in China. Series B, Chemistry, Life Sciences & Earth Sciences, 52(3), 332-337 (2009)
Fabrizio Carta et al.
Journal of medicinal chemistry, 56(11), 4691-4700 (2013-05-08)
Dithiocarbamates (DTCs) were recently discovered as carbonic anhydrase (CA, EC 4.2.1.1) inhibitors. A series of xanthates and a trithiocarbonate, structurally related to the DTCs, were prepared by reaction of alcohols/thiols with carbon disulfide in the presence of bases. These compounds
Protocols
Enzymatic Assay of Carbonic Anhydrase for Wilbur-Anderson Units (EC 4.2.1.1)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service