A86805
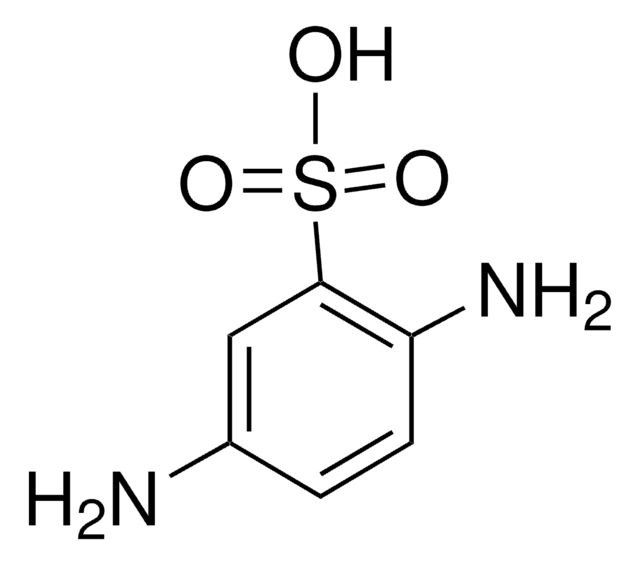
Aniline-2-sulfonic acid
95%
Synonym(s):
Orthanilic acid, 2-Aminobenzenesulfonic acid, o-Sulfanilic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
H2NC6H4SO3H
CAS Number:
Molecular Weight:
173.19
Beilstein:
1309204
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
form
solid
SMILES string
Nc1ccccc1S(O)(=O)=O
InChI
1S/C6H7NO3S/c7-5-3-1-2-4-6(5)11(8,9)10/h1-4H,7H2,(H,8,9,10)
InChI key
ZMCHBSMFKQYNKA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
J Mampel et al.
Microbiology (Reading, England), 145 ( Pt 11), 3255-3264 (1999-12-10)
Growth of Alcaligenes sp. strain O-1 with 2-aminobenzenesulfonate (ABS; orthanilate) as sole source of carbon and energy requires expression of the soluble, multicomponent 2-aminobenzenesulfonate 2,3-dioxygenase system (deaminating) (ABSDOS) which is plasmid-encoded. ABSDOS was separated by anion-exchange chromatography to yield a
Xin-Gui Li et al.
Journal of combinatorial chemistry, 8(2), 174-183 (2006-03-15)
A unique strategy for synthesis of narrowly distributed and inherently self-stabilized copolymer nanoparticles by a simple emulsifier-free polymerization from orthanilic acid and aniline was developed. The polymerization yield, electrical conductivity, size, and its distribution of the nanoparticles could be simultaneously
Inotropic activity of orthanilic and L-cysteic acid on isolated guinea-pig ventricular strips.
F Franconi et al.
Advances in experimental medicine and biology, 217, 159-165 (1987-01-01)
F Junker et al.
The Biochemical journal, 300 ( Pt 2), 429-436 (1994-06-01)
2-Aminobenzenesulphonic acid (2AS) is degraded by Alcaligenes sp. strain O-1 via a previously detected but unidentified intermediate. A mutant of strain O-1 was found to excrete this intermediate, which was isolated and identified by m.s., 1H- and 13C-n.m.r. as 3-sulphocatechol
Determination of nitrite ion and sulfanilic and orthanilic acids by differential pulse polarography.
S T Sulaiman
Analytical chemistry, 56(13), 2405-2407 (1984-11-01)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service