I12808
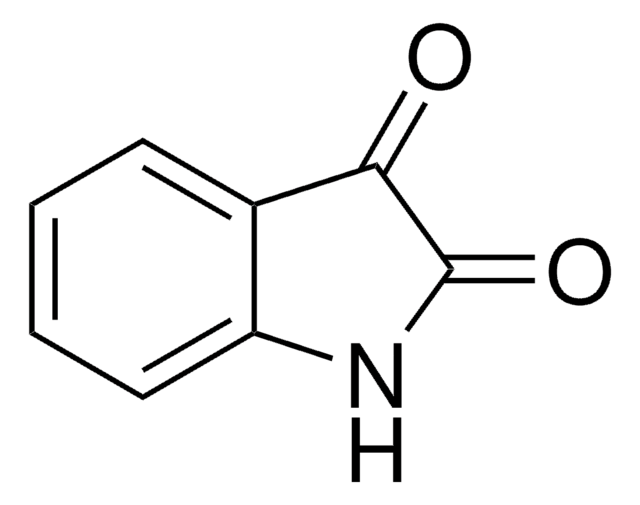
Isatoic anhydride
96%
Synonym(s):
3,1-Benzoxazine-2,4(1H)-dione, Anthranilic acid N-carboxylic acid anhydride
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C8H5NO3
CAS Number:
Molecular Weight:
163.13
Beilstein:
136786
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
vapor density
5.6 (vs air)
Quality Level
Assay
96%
mp
233 °C (dec.) (lit.)
SMILES string
O=C1Nc2ccccc2C(=O)O1
InChI
1S/C8H5NO3/c10-7-5-3-1-2-4-6(5)9-8(11)12-7/h1-4H,(H,9,11)
InChI key
TXJUTRJFNRYTHH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
586.4 °F - closed cup
Flash Point(C)
308 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
P S Gravett et al.
The International journal of biochemistry, 23(10), 1101-1110 (1991-01-01)
1. Esterase E-I from Bitis gabonica was inactivated with irreversible inhibitors which included studies with a water-soluble carbodiimide, an affinity labelling peptide and a mechanism-based inactivator. 2. The reaction with 1-ethyl-3(3-dimethylaminopropyl)-carbodiimide was biphasic and the dominant part followed saturation kinetics.
M H Gelb et al.
Journal of medicinal chemistry, 29(4), 585-589 (1986-04-01)
Derivatives of isatoic anhydride were prepared and tested as inhibitors of serine proteases. A number of isatoic anhydrides with positively charged substituents irreversibly inactivated several trypsin-like enzymes and preferentially inactivated trypsin over chymotrypsin. Further selectivity was obtained by introduction of
Asha M D'Souza et al.
Organic letters, 13(3), 486-489 (2010-12-24)
A nonstabilized azomethine ylide reacts with a wide range of substituted isatoic anhydrides to afford novel 1,3-benzodiazepin-5-one derivatives, which are generally isolated in high yield. The transformations involve 1,3-dipolar cycloaddition reactions of the ylide with the anhydrides to give transient
Zheng-Hui Guan et al.
Journal of the American Chemical Society, 134(42), 17490-17493 (2012-10-12)
A Pd-catalyzed regioselective C-H bond carbonylation of N-alkyl anilines for the synthesis of isatoic anhydrides has been developed. The key Pd-catalyst intermediate has been isolated and characterized. This novel Pd-catalyzed carbonylation reaction tolerates a wide range of functional groups and
Jonathan J Goodall et al.
Chembiochem : a European journal of chemical biology, 3(1), 68-75 (2007-06-27)
The acyl-enzyme formed upon acylation of alpha-chymotrypsin with isatoic anhydride has been characterised by infrared spectroscopy. Acylation at pH 7 to yield the 2-aminobenzoyl-enzyme is rapid (k = 5.57x 10(-2)s(-1)), while deacylation is much slower (k =3.7 x 10(-5)10(-2) (s-).
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service