P57204
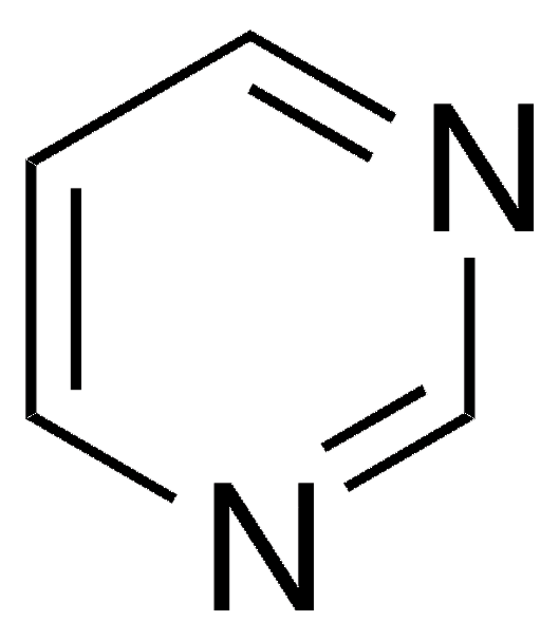
Pyridazine
98%
Synonym(s):
1,2-Diazabenzene, 1,2-Diazine, o-Diazine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C4H4N2
CAS Number:
Molecular Weight:
80.09
Beilstein:
103906
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
refractive index
n20/D 1.524 (lit.)
bp
208 °C (lit.)
mp
−8 °C (lit.)
density
1.103 g/mL at 25 °C (lit.)
SMILES string
c1ccnnc1
InChI
1S/C4H4N2/c1-2-4-6-5-3-1/h1-4H
InChI key
PBMFSQRYOILNGV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Pyridazine is a mono-basic 1,2-diazine compound, which is commonly prepared by the reaction of 1,4-dicarbonyls with hydrazines. Pyridazine ring is found in many herbicides like credazine, pyridatol and many pharmaceutical drugs like cefozopran, olaparib, talazoparib, and cadralazine.
Application
Pyridazine can be used:
- As a building block to synthesize N-phenyl-4-pyrazolo[1,5-b]pyridazin-3-yl-pyrimidin-2-amine derivatives as GSK-3 inhibitors.
- As a starting material in the synthesis of pharmacologically important pyrrolo[1,2-b]pyridazine derivatives.
- To prepare 1-(6-ethoxy-6-oxohexyl)pyridazin-1-ium bromide and 1-(2-bromoacetyl) pyridazinium bromide ionic liquids as corrosion inhibitors of steel under acidic conditions.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
185.0 °F - closed cup
Flash Point(C)
85 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Gernot Nuss et al.
Inorganic chemistry, 50(24), 12632-12640 (2011-11-19)
Reaction of potassium tris(mercapto-tert-butylpyridazinyl)borate K[Tn(tBu)] with copper(II) chloride in dichloromethane at room temperature led to the diamagnetic copper boratrane compound [Cu{B(Pn(tBu))(3)}Cl] (Pn = pyridazine-3-thionyl) (1) under activation of the B-H bond and formation of a Cu-B dative bond. In contrast
N-Phenyl-4-pyrazolo [1, 5-b] pyridazin-3-ylpyrimidin-2-amines as potent and selective inhibitors of glycogen synthase kinase 3 with good cellular efficacy
Tavares FX, et al.
Journal of Medicinal Chemistry, 47(19), 4716-4730 (2004)
Six membered heterocyclic compounds with two or more heteroatoms
Heterocyclic Chemistry (2010)
Masaomi Terajima et al.
European journal of pharmacology, 698(1-3), 455-462 (2012-11-28)
Given the key role p38 mitogen-activated protein kinase (MAPK) plays in inflammatory responses through the production of cytokines and inflammatory mediators, its inhibition is considered a promising therapeutic strategy for chronic inflammatory diseases such as rheumatoid arthritis, psoriasis, inflammatory bowel
Scott S Walker et al.
Antimicrobial agents and chemotherapy, 55(11), 5099-5106 (2011-08-17)
The echinocandins are a class of semisynthetic natural products that target β-1,3-glucan synthase (GS). Their proven clinical efficacy combined with minimal safety issues has made the echinocandins an important asset in the management of fungal infection in a variety of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)
