MX1123
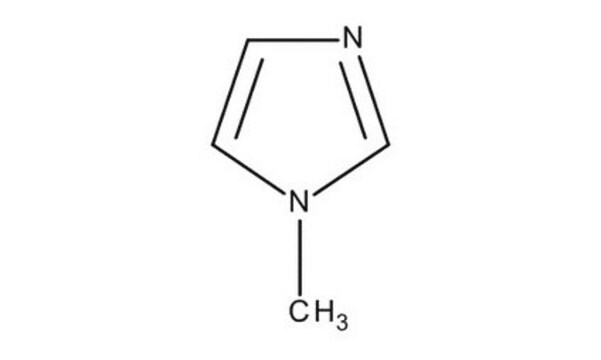
1-Methylimidazole
Synonym(s):
1-Methylimidazole, N-Methylimidazole, dna
About This Item
Recommended Products
vapor pressure
0.4 hPa ( 20 °C)
Quality Level
form
liquid
pH
9.5-10.5 (20 °C, 50 g/L in H2O)
bp
198 °C/1013 hPa
mp
-2 °C
transition temp
flash point 92 °C
shipped in
ambient
storage temp.
room temp
InChI
1S/C4H6N2/c1-6-3-2-5-4-6/h2-4H,1H3
InChI key
MCTWTZJPVLRJOU-UHFFFAOYSA-N
Related Categories
Application
- 4-amino-3,5-dicyano-6-arylphthalates via one-pot, a three-component reaction between arylidenemalononitriles, dialkyl acetylenedicarboxylates, and malononitrile.
- Aromatic and aliphatic carbamates from hydroxamic acids via the Lossen rearrangement.
- Tetrahydrobenzo[b]pyrans via three-component cyclocondensation reaction between aromatic aldehydes, malononitrile, and dimedone.
It can also be used as:
- A co-catalyst in copper-catalyzed, greener oxidation of alcohols to carbonyl compounds under aerobic conditions.
- A solvent/complexing agent in the preparation of CuInS2 n-type semiconductive particles.
- A co-additive in the synthesis of acid-sensitive epoxides by methyltrioxorhenium-catalyzed epoxidation of alkenes with H2O2.
Analysis Note
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Repr. 2 - Skin Corr. 1B
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 2
Flash Point(F)
197.6 °F - closed cup
Flash Point(C)
92 °C - closed cup
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service