136123
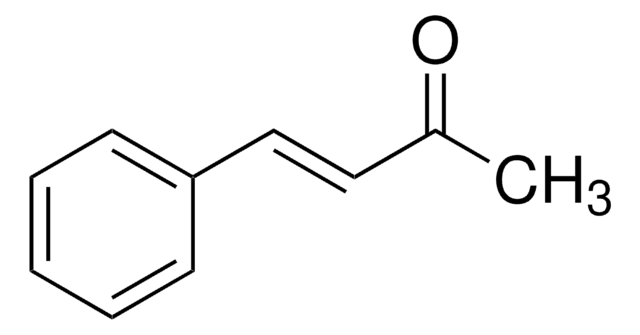
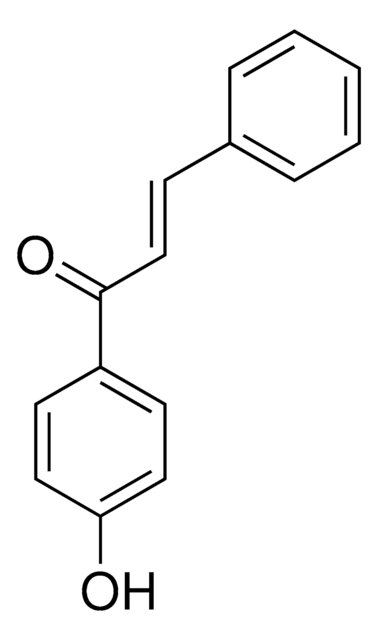
trans-Chalcone
97%
Synonym(s):
Benzylideneacetophenone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
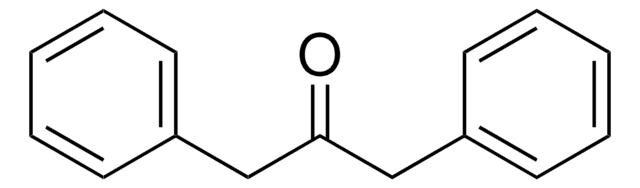
C6H5CH=CHCOC6H5
CAS Number:
Molecular Weight:
208.26
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
bp
208 °C/25 mmHg (lit.)
mp
55-57 °C (lit.)
functional group
ketone
phenyl
SMILES string
[H]\C(=C(\[H])C(=O)c1ccccc1)c2ccccc2
InChI
1S/C15H12O/c16-15(14-9-5-2-6-10-14)12-11-13-7-3-1-4-8-13/h1-12H/b12-11+
InChI key
DQFBYFPFKXHELB-VAWYXSNFSA-N
Gene Information
human ... IL1B(3553)
rat ... Ar(24208)
Related Categories
General description
trans-Chalcone is an open chain flavonoid that may prevent lung and forestomach cancer.
Application
trans-Chalcone was used in the synthesis of cis and trans diphenyl cyclopropane. It was also used in screening of surface adsorbed species of isobutybenzene and 4-isobutylacetophenone on bulk fosfotungstic Wells-Dawson acid (H6P2W18O62.xH2O).
Biochem/physiol Actions
trans-Chalcone exhibits antifungal activity against Trichophyton rubrum. It is inhibitor of fatty acid synthase and α-amylase. It induces programmed cell death due to reduced mitochondrial transmembrane potential in Arabidopsis thaliana roots.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Franco Chimenti et al.
Journal of medicinal chemistry, 52(9), 2818-2824 (2009-04-22)
A large series of substituted chalcones have been synthesized and tested in vitro for their ability to inhibit human monoamine oxidases A and B (hMAO-A and hMAO-B). While all the compounds showed hMAO-B selective activity in the micro- and nanomolar
P M Sivakumar et al.
Bioorganic & medicinal chemistry letters, 17(6), 1695-1700 (2007-02-06)
In order to develop relatively small molecules as antimycobacterial agents, twenty-five chalcones were synthesized, their activity was evaluated, and quantitative structure-activity relationship (QSAR) was developed. The synthesis was based on the Claisen-Schimdt scheme and the resultant compounds were tested for
Surface intermediate species of the 4-isobutylacetophenone adsorption-reaction over fosfotungstic Wells-Dawson heteropoly acid.
Matkovic SR, et al.
Latin American Applied Research, 39(2), 173-178 (2009)
Yi-Kun Chiang et al.
Journal of medicinal chemistry, 52(14), 4221-4233 (2009-06-11)
A pharmacophore model, Hypo1, was built on the basis of 21 training-set indole compounds with varying levels of antiproliferative activity. Hypo1 possessed important chemical features required for the inhibitors and demonstrated good predictive ability for biological activity, with high correlation
Wattenberg L.W., et al.
Cancer Letters, 15, 165-165 (1994)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service