405418
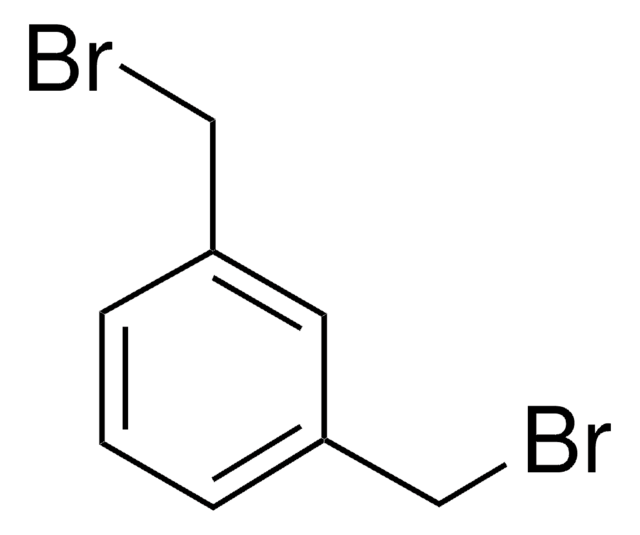
2,6-Bis(chloromethyl)pyridine
99%
Synonym(s):
α,α′-Dichloro-2,6-lutidine
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C7H7Cl2N
CAS Number:
Molecular Weight:
176.04
Beilstein:
116355
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
mp
73-78 °C (lit.)
functional group
chloro
SMILES string
ClCc1cccc(CCl)n1
InChI
1S/C7H7Cl2N/c8-4-6-2-1-3-7(5-9)10-6/h1-3H,4-5H2
InChI key
IWQNFYRJSVJWQA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2,6-Bis(chloromethyl)pyridine is a heterocyclic building block for the synthesis of a variety of pyridine derivatives. It coordinates with metal ions through N-atom to form complexes. The conformational flexibility of the bromomethyl arms makes it an ideal choice for the generation of macrocycles. 2,6-bis(chloromethyl)pyridine crystals are monoclinic with space group P21/c. Its synthesis has been reported. The FT-IR and FT-Raman spectra of 2,6-bis(chloromethyl)pyridine (BCMP) have been recorded in the regions 4000-400cm-1 and 3500-100cm-1, respectively.
Application
2,6-Bis(chloromethyl)pyridine may be used in the following studies:
- Synthesis of a sensitive fluorescent chemosensor for Hg2+, composed of two aminonaphthalimide fluorophores and a receptor of 2,6-bis(aminomethyl)pyridine.
- Preparation of carbene pincer ligands, required for the preparation of palladium pincer carbene complex.
- Synthesis of 2-(di-tert-butylphosphinomethyl)-6-diethylaminomethyl)pyridine, PNN ligand.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Xiangfeng Guo et al.
Journal of the American Chemical Society, 126(8), 2272-2273 (2004-02-26)
A selective and sensitive fluorescent chemosensor for Hg2+, which was composed of two aminonaphthalimide fluorophores and a receptor of 2,6-bis(aminomethyl)pyridine, was synthesized through the reaction of 2,6-bis(chloromethyl)pyridine and N-[2-(2-hydroxyethoxy)ethyl]-4-piperazino-1,8-naphthalimide. The chemosensor showed an about 17-fold increase in fluorescence quantum yield
Iron (II) complexes based on electron-rich, bulky PNN-and PNP-type ligands.
Zhang J, et al.
Inorgorganica Chimica Acta, 359(6), 1955-1960 (2006)
Tridentate carbene CCC and CNC pincer palladium (II) complexes: structure, fluxionality, and catalytic activity.
Grundemann S, et al.
Organometallics, 20(25), 5485-5488 (2001)
An efficient synthesis of 2,6-bis(chloromethyl)pyridine and a [5.5](2,6) pyridinophane disulfite.
Rezzonico B and Grignon-Dubois M.
J. Chem. Res. Synop., 4, 142-143 (1994)
V Balachandran et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 97, 1023-1032 (2012-08-29)
The FT-IR and FT-Raman spectra of 2,6-Bis(chloromethyl)pyridine (BCMP) have been recorded in the regions 4000-400 cm(-1) and 3500-100 cm(-1), respectively. The total energy calculations of BCMP were tried for the possible rotational isomers. The molecular structure, geometry optimization, vibrational frequencies
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service