A88255
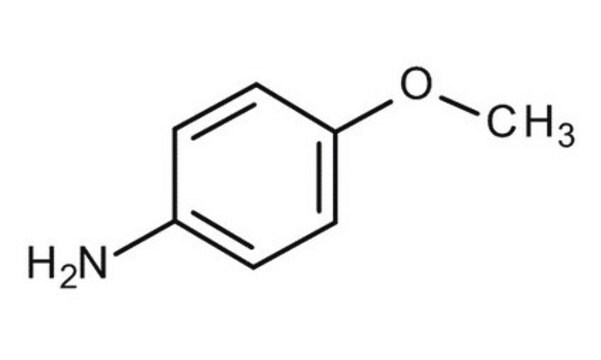
p-Anisidine
99%
Synonym(s):
4-Aminoanisole, 4-Methoxyaniline
About This Item
Recommended Products
Quality Level
Assay
99%
form
solid
autoignition temp.
959 °F
bp
240-243 °C (lit.)
mp
56-59 °C (lit.)
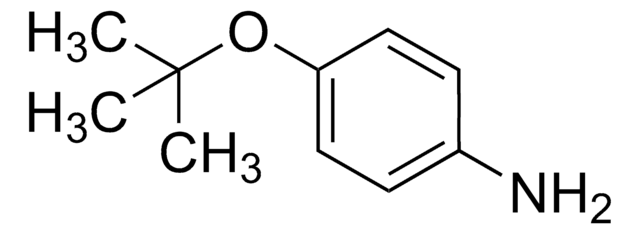
SMILES string
COc1ccc(N)cc1
InChI
1S/C7H9NO/c1-9-7-4-2-6(8)3-5-7/h2-5H,8H2,1H3
InChI key
BHAAPTBBJKJZER-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
p-Anisidine can be used:
- In the diastereoselective and enantioselective synthesis of CF3-substituted azoridines catalyzed by a chiral Bronsted acid.
- To prepare 4-organoselenium-quinolines through multi-component Povarov reaction with ethyl glyoxylate and ethynyl(phenyl)selane, catalyzed by Yb(OTf)3.
- As a starting material to synthesize 3-fluoro-6-methoxyquinoline in two steps.,
- To prepare N-PMP protected α-aminopropargylphosphonates by reacting with terminal alkynes and diethyl formylphosphonate hydrate using silver(I) triflate as a catalyst.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Carc. 1B - STOT RE 2
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
251.6 °F - closed cup
Flash Point(C)
122 °C - closed cup
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service