107794
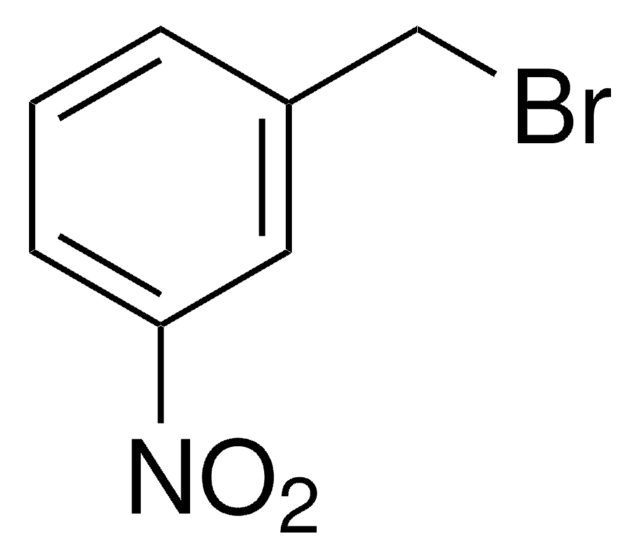
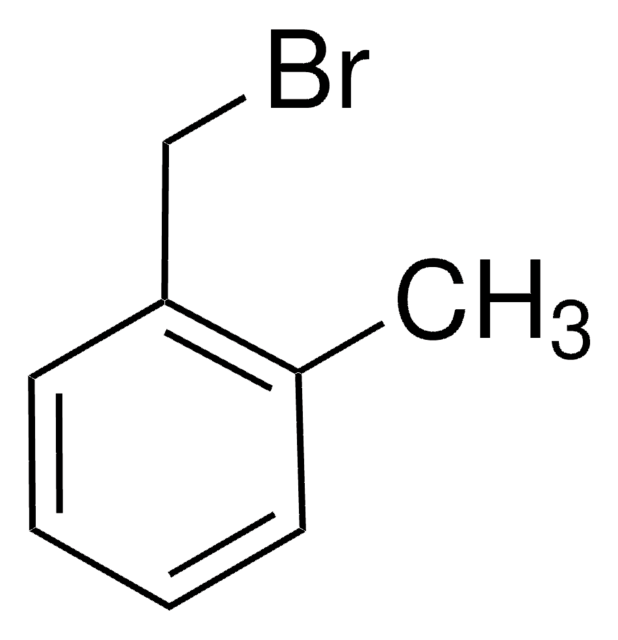
2-Nitrobenzyl bromide
98%
Synonym(s):
α-Bromo-2-nitrotoluene
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
O2NC6H4CH2Br
CAS Number:
Molecular Weight:
216.03
Beilstein:
638991
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
mp
44-46 °C (lit.)
functional group
bromo
nitro
SMILES string
[O-][N+](=O)c1ccccc1CBr
InChI
1S/C7H6BrNO2/c8-5-6-3-1-2-4-7(6)9(10)11/h1-4H,5H2
InChI key
HXBMIQJOSHZCFX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
2-Nitrobenzyl bromide was used for caging unprotected cysteine-containing or thiophosphorylated peptides in aqueous solution. It can be used in the synthesis of (R)- and (S)-3-amino-3,4-dihydro-1H-quinolin-2-one.
Biochem/physiol Actions
2-Nitrobenzyl bromide reacts with L-cysteine to form S-2-nitrobenzyl-cysteine which was used for modification of ultra-low-gelling-temperature (ULGT) agarose.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Xu Yang et al.
Polymers, 12(4) (2020-04-09)
Photoresponsive polymers have attracted increasing interest for a variety of applications. Here, we report a family of photoresponsive polypeptoid-based copolymer poly(ethylene glycol)-b-poly(N-(S-(o-nitrobenzyl)-thioethyl) glycine)-co-poly(N-(2-phenylethyl) glycine) (PEG-b-PNSN-co-PNPE) synthesized by the controlled ring-opening polymerization (ROP) technique. The key feature of the design is
Kyoung-Ho Park et al.
International journal of molecular sciences, 20(16) (2019-08-21)
A kinetic study was carried out on the solvolysis of o-nitrobenzyl bromide (o-isomer, 1) and p-nitrobenzyl bromide (p-isomer, 3), and o-nitrobenzoyl chloride (o-isomer, 2) in a wide range of solvents under various temperatures. In all of the solvents without aqueous
Ying Luo et al.
Nature materials, 3(4), 249-253 (2004-03-23)
Tissue engineering aims to replace, repair or regenerate tissue/organ function, by delivering signalling molecules and cells on a three-dimensional (3D) biomaterials scaffold that supports cell infiltration and tissue organization. To control cell behaviour and ultimately induce structural and functional tissue
P Pan et al.
FEBS letters, 405(1), 81-85 (1997-03-17)
Photoreleasable molecules are important in studies of various biological phenomena, especially cell signaling. Here we report a generally applicable approach for 'caging' unprotected cysteine-containing or thiophosphorylated peptides in aqueous solution with 2-nitrobenzyl bromides. Photolysis of the caged peptides was achieved
A practical synthesis of (R)-and (S)-3-amino-3, 4-dihydro-1H-quinolin-2-one.
Hulin B and Lopaze MG.
Tetrahedron Asymmetry, 15(12), 1957-1958 (2004)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service