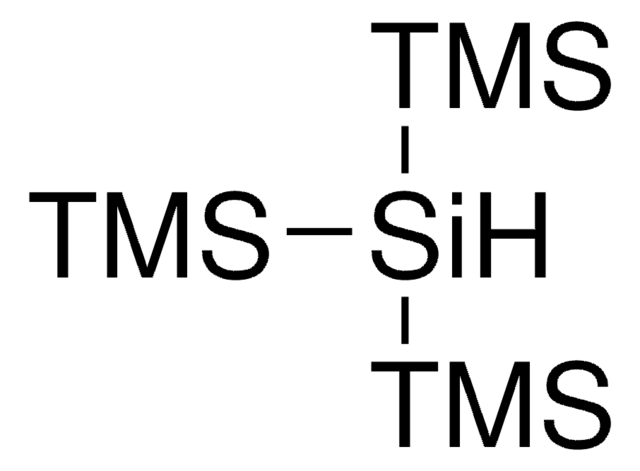736856
Tris(triethylsilyl)silane
Synonym(s):
1,1,1,3,3,3-Hexaethyl-2-(triethylsilyl)trisilane, 3,4,5-Trisilaheptane, 3,3,5,5-tetraethyl-4-(triethylsilyl)
About This Item
Recommended Products
form
liquid
Quality Level
reaction suitability
reagent type: reductant
refractive index
n20/D 1.526
density
0.887 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
CC[Si](CC)(CC)[SiH]([Si](CC)(CC)CC)[Si](CC)(CC)CC
InChI
1S/C18H46Si4/c1-10-20(11-2,12-3)19(21(13-4,14-5)15-6)22(16-7,17-8)18-9/h19H,10-18H2,1-9H3
InChI key
WNGZMQFMMHZKBG-UHFFFAOYSA-N
Application
It can be used as a reagent:
- In the radical coupling reaction to generate C-C bonds from alkyl-halogen compounds using iridium and nickel catalysts.
- To synthesize α-arylated product via cross-electrophile coupling reaction between α-chloro carbonyl and aryl bromide in the presence of nickel and iridium catalysts.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service