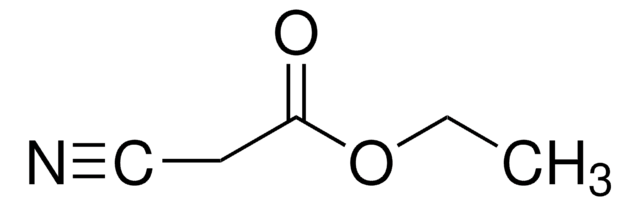
226319
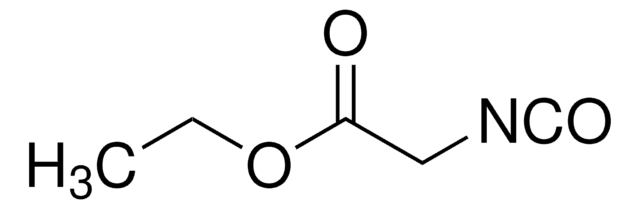
Ethyl isocyanoacetate
95%
동의어(들):
α-Isocyanoacetic acid ethyl ester, (Ethoxycarbonyl)methyl isonitrile, 2-Ethoxy-2-oxoethyl isocyanide, 2-Ethyloxycarbonylmethyl isocyanide, Ethoxycarbonylmethyl isocyanide, Ethyl α-isocyanoacetate, Ethyl 2-isocyanoacetate, Isocyanoacetic acid ethyl ester
About This Item
추천 제품
분석
95%
양식
liquid
refractive index
n20/D 1.418 (lit.)
bp
194-196 °C (lit.)
density
1.035 g/mL at 25 °C (lit.)
작용기
amine
ester
isonitrile
저장 온도
2-8°C
SMILES string
CCOC(=O)C[N+]#[C-]
InChI
1S/C5H7NO2/c1-3-8-5(7)4-6-2/h3-4H2,1H3
InChI key
FPULFENIJDPZBX-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
183.2 °F - closed cup
Flash Point (°C)
84 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.


![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)