모든 사진(3)
About This Item
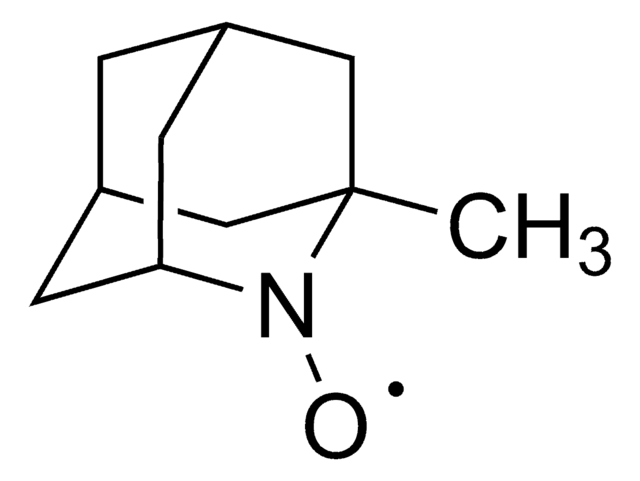
실험식(Hill 표기법):
C9H14NO
CAS Number:
Molecular Weight:
152.21
MDL number:
UNSPSC 코드:
12352000
PubChem Substance ID:
NACRES:
NA.22
가격 및 재고 정보를 현재 이용할 수 없음
추천 제품
분석
90%
양식
powder
반응 적합성
reagent type: oxidant
mp
182-189 °C (D)
저장 온도
2-8°C
SMILES string
[O]N1[C@@H]2C[C@H]3C[C@@H](C2)C[C@@H]1C3
InChI
1S/C9H14NO/c11-10-8-2-6-1-7(4-8)5-9(10)3-6/h6-9H,1-5H2/t6-,7+,8-,9+
InChI key
BCJCJALHNXSXKE-SPJNRGJMSA-N
일반 설명
2-Azaadamantane-N-oxyl (AZADO), a stable nitroxyl radical, is widely employed as catalyst for the oxidation of alcohols.[1]
이미 열람한 고객
Ming Zhang et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 21(10), 3937-3941 (2015-01-22)
An increased supply of scarce or inaccessible natural products is essential for the development of more sophisticated pharmaceutical agents and biological tools, and thus the development of atom-economical, step-economical and scalable processes to access these natural products is in high
Masatoshi Shibuya et al.
Journal of the American Chemical Society, 128(26), 8412-8413 (2006-06-29)
Development of a stable nitroxyl radical class of catalysts, 2-azaadamantane N-oxyl (AZADO) and 1-Me-AZADO, for highly efficient oxidation of alcohols is described. AZADO and 1-Me-AZADO exhibit superior catalytic proficiency to TEMPO, converting various sterically hindered alcohols to the corresponding carbonyl
Takefumi Kuranaga et al.
Journal of the American Chemical Society, 135(14), 5467-5474 (2013-03-19)
Here we report the first total synthesis and the complete stereochemical assignment of yaku'amide A. Yaku'amide A (1) was isolated from a sponge Ceratopsion sp. as an extremely potent cytotoxin. Its structure was determined except for the C4-stereochemistry in the
Takuya Isogai et al.
Biomacromolecules, 11(6), 1593-1599 (2010-05-18)
Curdlan, amylodextrin, and regenerated cellulose fiber were subjected to electromediated oxidation with a 4-acetamido-TEMPO catalyst in a buffer at pH 6.8 without NaClO or NaClO(2). More than 90% of the C6 primary hydroxyls of Curdlan and amylodextrin were converted to
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.![9-Azabicyclo[3.3.1]nonane N-oxyl 95%](/deepweb/assets/sigmaaldrich/product/structures/287/155/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf/640/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf.png)