T84654
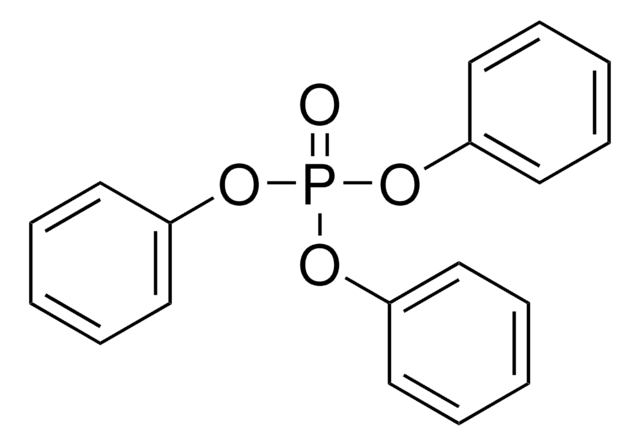
Triphenyl phosphite
97%
Synonym(s):
(PhO)3P, P(OPh)3, Triphenoxyphosphine
About This Item
Recommended Products
vapor density
10.7 (vs air)
vapor pressure
5 mmHg ( 205 °C)
Assay
97%
reaction suitability
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
reaction type: Alkylations
reagent type: ligand
reaction type: Cycloadditions
reagent type: ligand
reaction type: Heck Reaction
reagent type: ligand
reaction type: Olefinations
reagent type: ligand
reaction type: Stille Coupling
reagent type: ligand
reaction type: Wittig Reaction
refractive index
n20/D 1.59 (lit.)
bp
360 °C (lit.)
mp
22-24 °C (lit.)
density
1.184 g/mL at 25 °C (lit.)
SMILES string
O(P(Oc1ccccc1)Oc2ccccc2)c3ccccc3
InChI
1S/C18H15O3P/c1-4-10-16(11-5-1)19-22(20-17-12-6-2-7-13-17)21-18-14-8-3-9-15-18/h1-15H
InChI key
HVLLSGMXQDNUAL-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- As a source of phosphorus and as a ligand for the synthesis of transition metal phosphide nanoparticles via heating-up process.
- To convert alcohols to alkyl halides.
- As a peptide coupling agent.
- As a low-temperature source of singlet oxygen after forming an adduct with ozone.
- To synthesize bromotriphenoxyphosphonium bromide, a brominating agent, by reacting with bromine.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Skin Irrit. 2 - Skin Sens. 1 - STOT RE 2
Target Organs
Nervous system
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
410.0 °F
Flash Point(C)
210 °C
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
The Catalexis platform enhances catalysis by digitally optimizing catalyst selection to identify the most effective phosphine ligands for cross-coupling reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service