E6412
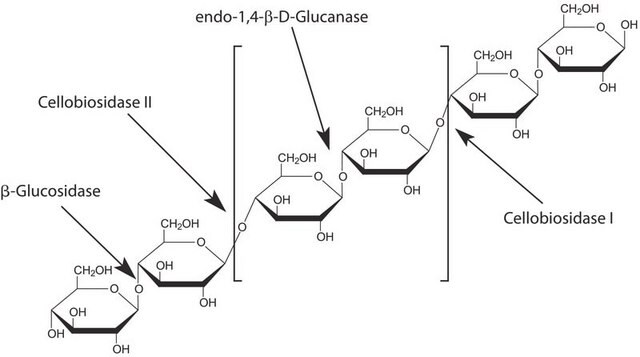
Cellobiohydrolase I from Hypocrea jecorina
0.13 U/mg, recombinant, expressed in corn
Synonym(s):
Cel7A, Cellobiosidase, Cellulase
About This Item
Recommended Products
recombinant
expressed in corn
Quality Level
form
liquid
specific activity
0.13 U/mg
greener alternative product characteristics
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
greener alternative category
shipped in
dry ice
storage temp.
−20°C
General description
Application
Biochem/physiol Actions
Unit Definition
Physical form
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Uncover more about glycosaminoglycans and proteoglycans including the structure of glycosaminoglycans (GAGs), the different types of GAGs, and their functions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service