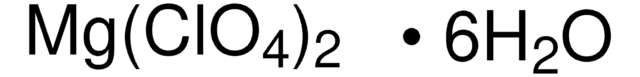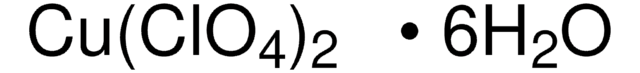모든 사진(2)
About This Item
Linear Formula:
Mg(ClO4)2
CAS Number:
Molecular Weight:
223.21
EC Number:
MDL number:
UNSPSC 코드:
12352300
PubChem Substance ID:
NACRES:
NB.24
추천 제품
Grade
ACS reagent
양식
flakes
powder, chunks or granules
반응 적합성
reagent type: oxidant
농도
>10% Mg (EDTA titration)
불순물
≤0.005 meq/g Titr. free acid
≤0.025 meq/g Titr. base
손실
≤8% loss on drying
적합성
passes test for moisture absorption
SMILES string
[Mg++].[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O
InChI
1S/2ClHO4.Mg/c2*2-1(3,4)5;/h2*(H,2,3,4,5);/q;;+2/p-2
InChI key
MPCRDALPQLDDFX-UHFFFAOYSA-L
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Magnesium perchlorate (Mg(ClO4)2) is widely used as drying agent for gases. It can remove water from gases (with no organic contaminants) at the rate of 0.001mgwater/l.
애플리케이션
Magnesium perchlorate (Mg(ClO4)2) may be employed as a catalyst in the following studies:
- Preparation of α-aminophosphonates.
- Enantioselective Diels-Alder reaction between cyclopentadiene and 3-acryloyl-1,3-oxazolin-2-one.
- Preparation of imines and phenylhydrazones.
- Protection of alcohols in the form of t-butyl ethers.
Magnesium perchlorate may be used as a catalyst in the synthesis of the following compounds:
- α-Aminophosphonates via three-component reaction between an amine, an aldehyde or a ketone and a di-/trialkyl phosphite.
- Imines and phenylhydrazones by the condensation of carbonyl compounds with amines and phenylhydrazine.
- Knoevenagel adducts via Knoevenagel condensation between β-diketones and aliphatic or aromatic aldehydes.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Ox. Sol. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
5.1A - Strongly oxidizing hazardous materials
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
Magnesium perchlorate as efficient Lewis acid for the Knoevenagel condensation between ?-diketones and aldehydes.
Bartoli G, et al.
Tetrahedron Letters, 49(16), 2555-2557 (2008)
The first enantioselective synthesis of both Diels-Alder enantiomers with the same bis (oxazoline)-magnesium perchlorate chiral catalyst.
Desimoni G, et al.
Tetrahedron Letters, 37(17), 3027-3030 (1996)
Eagleson M.
Concise Encyclopedia Chemistry, 343-343 (1994)
Magnesium perchlorate as an efficient catalyst for the synthesis of imines and phenylhydrazones.
Chakraborti AK, et al.
Tetrahedron Letters, 45(41), 7641-7644 (2004)
Giuseppe Bartoli et al.
Organic letters, 7(3), 427-430 (2005-01-28)
[reaction: see text] A new mild method for protecting alcohols as t-butyl ethers is reported. The reaction proceeds with Mg(ClO4)2 and Boc2O and shows general applicability. The deprotection of t-butyl ethers has also been revisited. Preliminary results indicate the CeCl3
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.