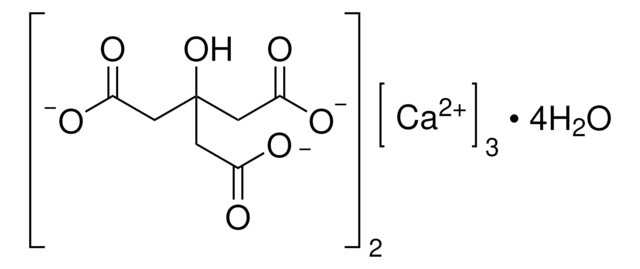
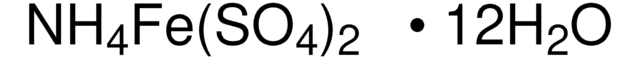추천 제품
Grade
ACS reagent
Quality Level
분석
≥99.0%
양식
powder
불순물
≤0.005% insolubles
≤0.035 meq/g Titr. acid
음이온 미량물
chloride (Cl-): ≤0.001%
sulfate (SO42-): ≤0.01%
양이온 미량물
Ba: ≤0.01%
Fe: ≤0.001%
K: ≤0.01%
Mg: ≤0.05%
Na: ≤0.02%
Sr: ≤0.05%
heavy metals (as Pb): ≤0.005%
적합성
passes test for acidity or alkalinity
SMILES string
[H]O[H].CC(=O)O[Ca]OC(C)=O
InChI
1S/2C2H4O2.Ca.H2O/c2*1-2(3)4;;/h2*1H3,(H,3,4);;1H2/q;;+2;/p-2
InChI key
XQKKWWCELHKGKB-UHFFFAOYSA-L
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Calcium acetate monohydrate is calcium salt of acetic acid. Crystals study reveals the structure to be triclinic. Its solubility in aqueous solution over a range of temperature has been studied.
애플리케이션
Calcium acetate monohydrate may be used in the following processes:
- As a calcium source in the preparation of calcium hydroxyapatite by sol–gel synthesis.
- As a precursor in the synthesis of calcium oxide sorbent used in capturing CO2.
- As a precursor salt in the synthesis of CaO/γ-Al2O3 ( calcium oxide/ aluminium oxide) heterogeneous base catalysts.
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
The synthesis of CaO loaded onto Al2O3 from calcium acetate and its application in transesterification of the sunflower oil.
Marinkovic DM, et al.
Advanced technologies, 4(1) , 26-32 (2015)
Calcium hydroxyapatite, Ca10(PO4)6(OH)2 ceramics prepared by aqueous sol-gel processing.
Bogdanoviciene I, et al.
Materials Research Bulletin, 41(9), 1754-1762 (2006)
Ash M and Ash I.
Handbook of Preservatives, 309-309 (2004)
Solubilities of calcium acetates in the temperature range 0-100?C.
Saury C, et al.
Journal of Chemical and Engineering Data, 38(1), 56-59 (1993)
Structure of calcium acetate monohydrate, Ca(C2H3O2) 2.H2O.
Klop EA, et al.
Acta Crystallographica Section C, Crystal Structure Communications, 40(1), 51-53 (1984)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.