추천 제품
분석
≥90%
저장 온도
−20°C
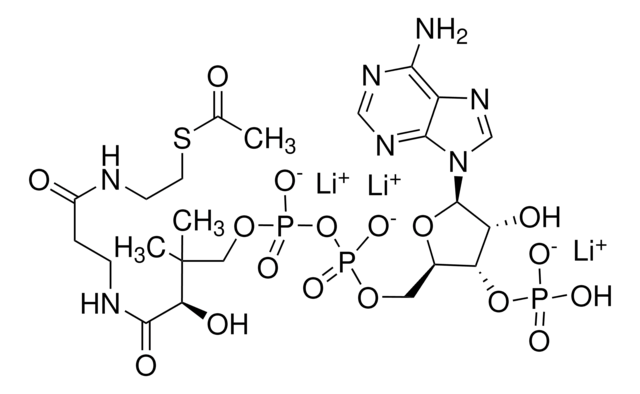
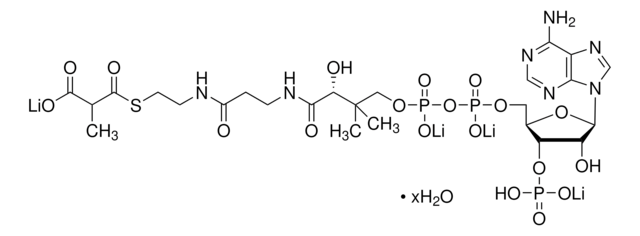
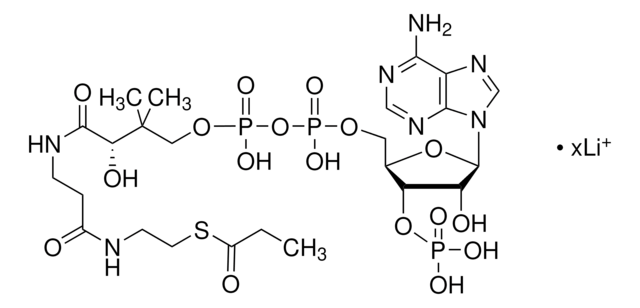
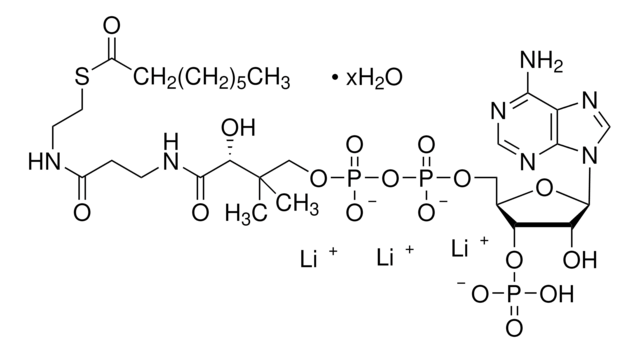
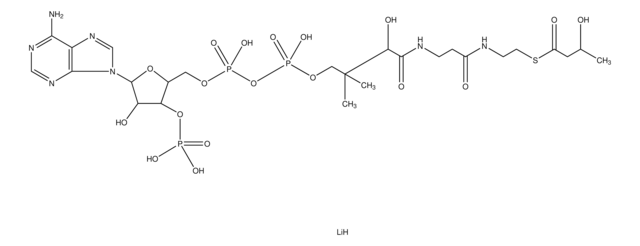
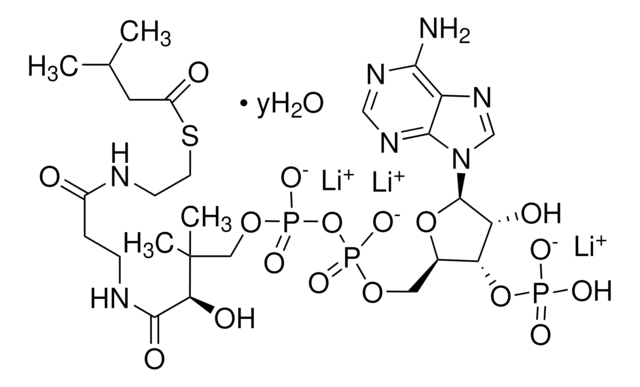
SMILES string
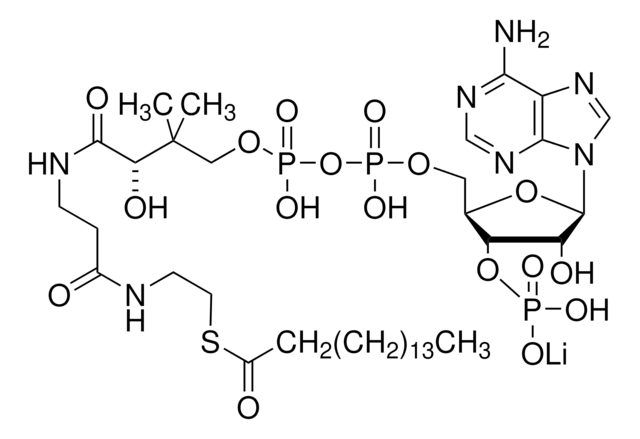
[Li].CC(C)(COP(O)(=O)OP(O)(=O)OCC1OC(C(O)C1OP(O)(O)=O)n2cnc3c(N)ncnc23)C(O)C(=O)NCCC(=O)NCCSC(=O)CCCC(O)=O
InChI
1S/C26H42N7O19P3S/c1-26(2,21(39)24(40)29-7-6-15(34)28-8-9-56-17(37)5-3-4-16(35)36)11-49-55(46,47)52-54(44,45)48-10-14-20(51-53(41,42)43)19(38)25(50-14)33-13-32-18-22(27)30-12-31-23(18)33/h12-14,19-21,25,38-39H,3-11H2,1-2H3,(H,28,34)(H,29,40)(H,35,36)(H,44,45)(H,46,47)(H2,27,30,31)(H2,41,42,43)/t14-,19-,20-,21?,25-/m1/s1
InChI key
SYKWLIJQEHRDNH-KRPIADGTSA-N
일반 설명
Glutaryl coenzyme A (Glutaryl CoA) is an intermediate in the mitochondrial oxidation of lysine, hydroxylysine and tryptophan.
애플리케이션
Glutaryl coenzyme A lithium salt has been used:
- in comparative study of acylomes of β-Hydroxy β-methylglutaryl-CoA (HMG-CoA) and glutaryl-CoA by quantitative proteomics
- as a component of the assay buffer for in vitro biosensor activity experiments for FapR-NLuc proteins
- to test its effect on pyruvate kinase activity inhibition in a549 lysate
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
Structural basis for promoting and preventing decarboxylation in glutaryl-coenzyme a dehydrogenases.
Simon Wischgoll et al.
Biochemistry, 49(25), 5350-5357 (2010-05-22)
Glutaryl-coenzyme A dehydrogenases (GDHs) involved in amino acid degradation were thought to catalyze both the dehydrogenation and decarboxylation of glutaryl-coenzyme A to crotonyl-coenzyme A and CO(2). Recently, a structurally related but nondecarboxylating, glutaconyl-coenzyme A-forming GDH was characterized in the obligately
Rhushikesh A Kulkarni et al.
Cell chemical biology, 24(2), 231-242 (2017-02-07)
Non-enzymatic protein modification driven by thioester reactivity is thought to play a major role in the establishment of cellular lysine acylation. However, the specific protein targets of this process are largely unknown. Here we report an experimental strategy to investigate
Kinetic mechanism of glutaryl-CoA dehydrogenase
Rao K S, et al.
Biochemistry, 45(51), 15853-15861 (2006)
Genetically Encoded FapR-NLuc as a Biosensor to Determine Malonyl-CoA in Situ at Subcellular Scales.
Yipeng Du et al.
Bioconjugate chemistry, 30(3), 826-832 (2019-01-11)
Malonyl-CoA is one of the key metabolic intermediates in fatty acid metabolism as well as a key player in protein post-translational modifications. Detection of malonyl-CoA in live cells is challenging because of the lack of effective measuring tools. Here we
A Class of Reactive Acyl-CoA Species Reveals the Non-enzymatic Origins of Protein Acylation
Wagner GR, et al.
Cell Metabolism, 25(4), 823-837 (2017)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.