H9384
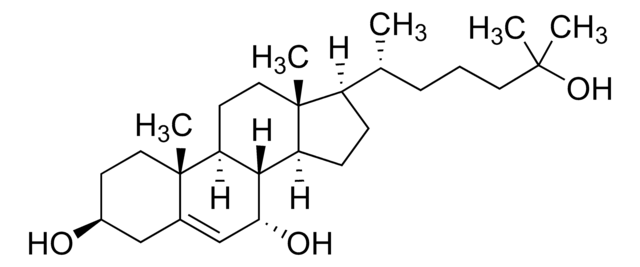
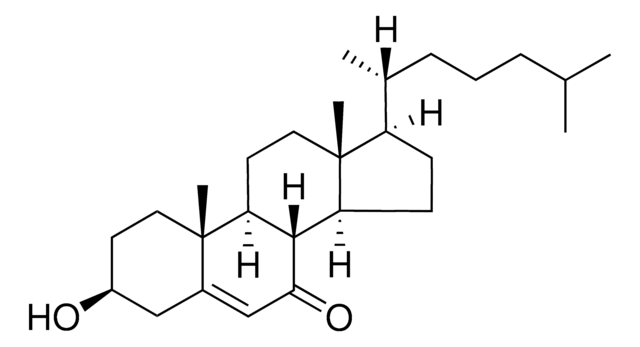
22(R)-Hydroxycholesterol
≥98%
동의어(들):
22α-Hydroxycholesterol, 5-Cholestene-3β,22(R)-diol, 5-Cholestene-3β,22[R]-diol
로그인조직 및 계약 가격 보기
크기 선택
모든 사진(1)
크기 선택
보기 변경
About This Item
실험식(Hill 표기법):
C27H46O2
CAS Number:
Molecular Weight:
402.65
MDL number:
UNSPSC 코드:
12352211
eCl@ss:
39023139
PubChem Substance ID:
NACRES:
NA.77
추천 제품
Quality Level
분석
≥98%
양식
powder
배송 상태
ambient
저장 온도
room temp
SMILES string
[H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)[C@@]1([H])CC[C@]4(C)[C@H](CC[C@@]24[H])[C@H](C)[C@H](O)CCC(C)C
InChI
1S/C27H46O2/c1-17(2)6-11-25(29)18(3)22-9-10-23-21-8-7-19-16-20(28)12-14-26(19,4)24(21)13-15-27(22,23)5/h7,17-18,20-25,28-29H,6,8-16H2,1-5H3/t18-,20-,21-,22+,23-,24-,25+,26-,27+/m0/s1
InChI key
RZPAXNJLEKLXNO-GFKLAVDKSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
Bovine aortic endothelial cells were treated with 22(R)-Hydroxycholesterol to study the effects on production of free radicals and in studies related to fatty acid metabolism.
생화학적/생리학적 작용
22(R)-Hydroxycholesterol is an intermediate of the pregnenolone synthesis pathway from cholesterol. It reported has neuroprotective properties and protects the neurons against β-amyloid-induced cell death. 22(R)-Hydroxycholesterol acts as the ligand of liver X receptors that act as sensors of sterol concentration and regulates the fatty acid metabolism.
제조 메모
22(R)-Hydroxycholesterol yield clear, colorless solution in chloroform.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
A Chawla et al.
Science (New York, N.Y.), 294(5548), 1866-1870 (2001-12-01)
Cholesterol, fatty acids, fat-soluble vitamins, and other lipids present in our diets are not only nutritionally important but serve as precursors for ligands that bind to receptors in the nucleus. To become biologically active, these lipids must first be absorbed
Lourdes Cruz-Garcia et al.
Comparative biochemistry and physiology. Part A, Molecular & integrative physiology, 160(2), 125-136 (2011-06-04)
The liver X receptor (LXR) has recently been described in salmonids. In mammals, this receptor is already known as a transcriptional factor that regulates diverse aspects of cholesterol, fatty acid and carbohydrate metabolism in various tissues, including muscle. Here we
Cynthia Hong et al.
Journal of lipid research, 52(3), 531-539 (2010-12-29)
Ligand activation of liver X receptors (LXRs) has been shown to impact both lipid metabolism and inflammation. One complicating factor in studies utilizing synthetic LXR agonists is the potential for pharmacologic and receptor-independent effects. Here, we describe an LXR gain-of-function
Linara Gabitova et al.
Cell reports, 12(11), 1927-1938 (2015-09-08)
Meiosis-activating sterols (MAS) are substrates of SC4MOL and NSDHL in the cholesterol pathway and are important for normal organismal development. Oncogenic transformation by epidermal growth factor receptor (EGFR) or RAS increases the demand for cholesterol, suggesting a possibility for metabolic
Nina Hallmark et al.
Environmental health perspectives, 115(3), 390-396 (2007-04-14)
Certain phthalates can impair Leydig cell distribution and steroidogenesis in the fetal rat in utero, but it is unknown whether similar effects might occur in the human. Our aim in this study was to investigate the effects of di(n-butyl) phthalate
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.