A71301
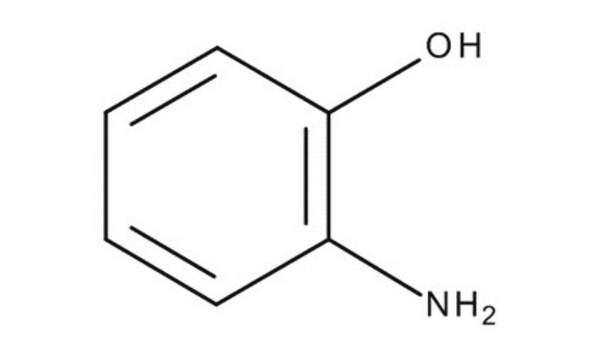
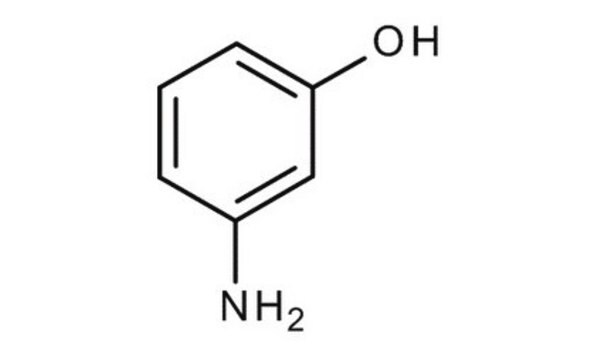
2-Aminophenol
99%
동의어(들):
(2-Hydroxyphenyl)amine, 1-Hydroxy-2-aminobenzene, 2-Amino-1-hydroxybenzene, 2-Aminophenol, 2-Aminophenyl alcohol, 2-Hydroxyaniline, 2-Hydroxybenzenamine, o-Aminohydroxybenzene, o-Hydroxyaniline, o-Hydroxyphenylamine
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
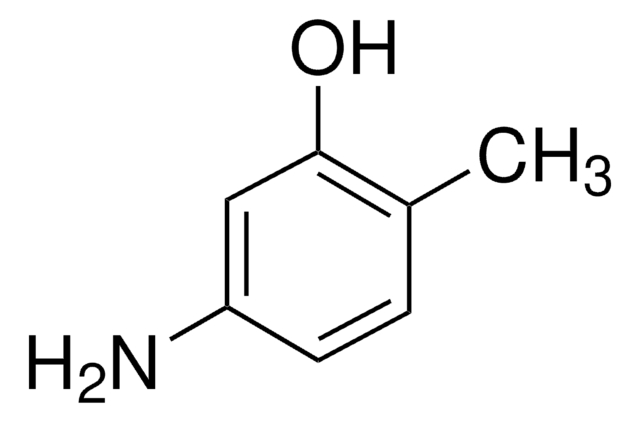
H2NC6H4OH
CAS Number:
Molecular Weight:
109.13
Beilstein:
606075
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
99%
양식
powder
mp
170-175 °C (lit.)
SMILES string
Nc1ccccc1O
InChI
1S/C6H7NO/c7-5-3-1-2-4-6(5)8/h1-4,8H,7H2
InChI key
CDAWCLOXVUBKRW-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
애플리케이션
2-Aminophenol can be used in the synthesis of:
- 2-Oxazolidinone derivatives by reacting with β-aminoalcohols in presence of Pd/C-I2 as a catalyst via oxidative cyclocarbonylation.
- Schiff base transition metal(II) complexes with salicylidene-4-aminoantipyrine.
- 2-Arylbenzoxazoles with aldehydes catalyzed by activated carbon in presence of oxygen atmosphere.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Muta. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 2
Flash Point (°F)
334.4 °F - closed cup
Flash Point (°C)
168 °C - closed cup
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Synthesis, spectroscopic characterization, redox, and biological screening studies of some Schiff base transition metal (II) complexes derived from salicylidene-4-aminoantipyrine and 2-aminophenol/2-aminothiophenol.
Raman N, et al.
Synth. React. Inorg. Met.-Org. Chem. , 31(7), 1249-1270 (2001)
Synthesis of 2-oxazolidinone catalyzed by palladium on charcoal: a novel and highly effective heterogeneous catalytic system for oxidative cyclocarbonylation of β -aminoalcohols and 2-aminophenol.
Li F and Xia C
J. Catal., 227(2), 542-546 (2004)
Yoshinori Endo et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 18(43), 13609-13613 (2012-09-13)
Aerobic oxidation: in a biomimetic approach, a mixture of redox catalysts forms couples that effect the aerobic oxidation of a mixture of benzylamine and 2-aminophenol derivatives to give the corresponding benzoxazoles. This biomimetic oxidation proceeds smoothly under mild conditions and
Michael M Bittner et al.
Journal of the American Chemical Society, 134(12), 5460-5463 (2012-03-16)
The oxidative ring cleavage of aromatic substrates by nonheme Fe dioxygenases is thought to involve formation of a ferrous-(substrate radical) intermediate. Here we describe the synthesis of the trigonal-bipyramdial complex Fe((Ph2)Tp)(ISQ(tBu)) (2), the first synthetic example of an iron(II) center
Ericsson D Coy et al.
Organic letters, 12(9), 1976-1979 (2010-03-27)
An efficient and facile synthesis of trans-dihydrobenzofurans has been accomplished from o-aminophenols and phenylpropenes via a novel (one-pot) diastereoselective Pd-catalyzed oxyarylation reaction. The development and optimization of this method is described.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.