추천 제품
Quality Level
분석
≥98% (HPLC)
양식
powder
저장 조건
desiccated
색상
faintly yellow to dark yellow
solubility
H2O: ≥5 mg/mL
저장 온도
2-8°C
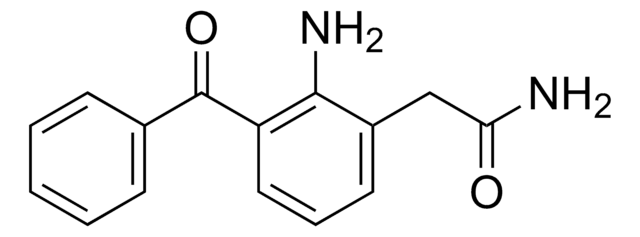
SMILES string
[Na+].Nc1c(CC([O-])=O)cccc1C(=O)c2ccc(Br)cc2
InChI
1S/C15H12BrNO3.Na/c16-11-6-4-9(5-7-11)15(20)12-3-1-2-10(14(12)17)8-13(18)19;/h1-7H,8,17H2,(H,18,19);/q;+1/p-1
InChI key
HZFGMQJYAFHESD-UHFFFAOYSA-M
유전자 정보
human ... PTGS1(5742) , PTGS2(5743)
애플리케이션
Bromfenac sodium has been used:
- to study its ability to bind to melanin
- in the synthesis of bromfenac indolinone standard
- to analyze its permeability in porcine conjunctiva
생화학적/생리학적 작용
Bromfenac exhibits antipyretic and prostaglandin synthetase inhibiting properties. It has therapeutic properties against the reduction of ocular pain and inflammation in postoperative cataract patients. Bromfenac acts as an effective agent against allergic conjunctivitis. It has the potential to treat acute muscle pain, osteoarthritis, and rheumatoid arthritis.
Bromfenac is a nonsteroidal anti inflammatory drug (NSAID) that inhibits both COX1 and COX2. It is used as an opthalmic analgesic.
Bromfenac is a nonsteroidal anti inflammatory drug (NSAID); COX1 and COX2 inhibitor; opthalmic analgesic.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Oral
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
Thomas R Walters et al.
Ophthalmology, 121(1), 25-33 (2013-09-12)
To evaluate the efficacy and ocular safety of bromfenac ophthalmic solution 0.07% (Prolensa) dosed once daily for the treatment of ocular inflammation and pain in subjects who underwent cataract surgery with posterior chamber intraocular lens implantation. Two phase 3, randomized
Eiichi Uchio et al.
Ophthalmologica. Journal international d'ophtalmologie. International journal of ophthalmology. Zeitschrift fur Augenheilkunde, 221(3), 153-158 (2007-04-19)
We evaluated the efficacy and safety of long-term management of patients with vernal keratoconjunctivitis (VKC) with bromfenac sodium eye drops in combination with corticosteroids and anti-allergic eye drops. Twenty-two patients with VKC were randomly assigned to receive two test eye
Sandrine A Zweifel et al.
Retina (Philadelphia, Pa.), 29(10), 1527-1531 (2009-11-10)
The purpose of this study was to assess the efficacy of topical bromfenac (0.09%) as an adjunctive therapy for patients with neovascular age-related macular degeneration demonstrating persistent exudation despite monthly intravitreal antivascular endothelial growth factor therapy. Twenty-one patients (22 eyes)
Christina Flaxel et al.
Retina (Philadelphia, Pa.), 32(3), 417-423 (2011-08-25)
To evaluate whether bromfenac eyedrops and ranibizumab intravitreal injections would provide added efficacy over ranibizumab alone. This was a single-site, multiinvestigator, prospective, open-label, interventional, Phase II study of patients with new or recurrent exudative/neovascular age-related macular degeneration. Thirty eyes were
Jeffrey S Heier et al.
Retina (Philadelphia, Pa.), 29(9), 1310-1313 (2009-11-26)
To assess vitreous concentrations of nonsteroidal antiinflammatory drugs (NSAIDs) and prostaglandin E(2) in patients treated with NSAIDs before vitrectomy. This was an investigator-masked, randomized, multicenter study. Patients received ketorolac 0.4% 4 times a day, bromfenac 0.09% 2 times a day
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.