추천 제품
분석
≥98% (HPLC)
양식
powder
저장 조건
desiccated
색상
white to brown
solubility
DMSO: 10 mg/mL, clear
저장 온도
−20°C
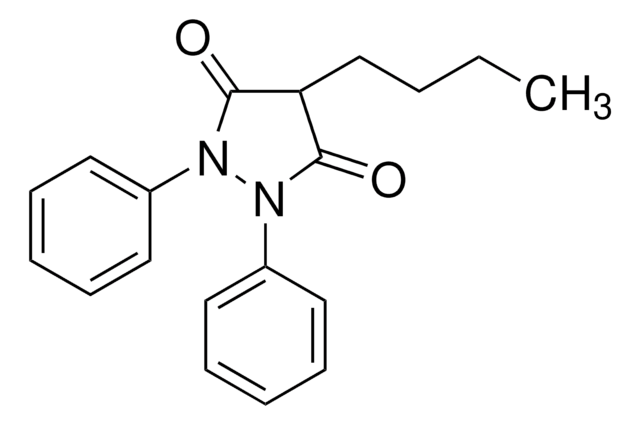
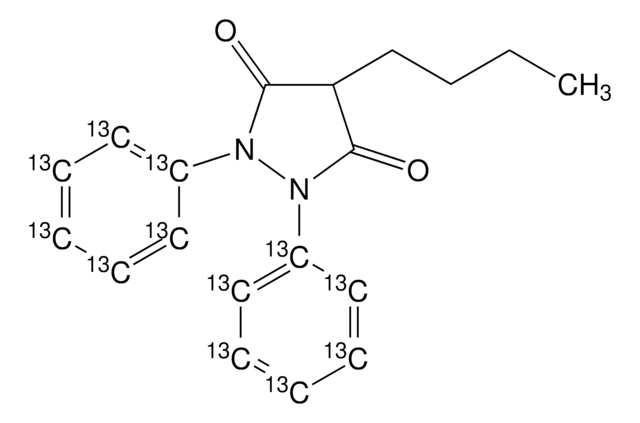
SMILES string
N2(N(C(=O)C(C2=O)CCCC)c3ccccc3)c1ccc(cc1)O
InChI
1S/C19H20N2O3/c1-2-3-9-17-18(23)20(14-7-5-4-6-8-14)21(19(17)24)15-10-12-16(22)13-11-15/h4-8,10-13,17,22H,2-3,9H2,1H3
InChI key
HFHZKZSRXITVMK-UHFFFAOYSA-N
일반 설명
Oxyphenbutazone is a derivative compound of phenylbutazone.
생화학적/생리학적 작용
Oxyphenbutazone is a non-steroid anti inflammatory; anti Mycobacterium tuberculosis agent.
Oxyphenbutazone is a non-steroid anti inflammatory; anti Mycobacterium tuberculosis agent. Oxyphenbutazone is known to cause inflammatory effects on tissues. Oxyphenbutazone, as a drug, decreases cellular exudates, without involving the pituitary-adrenal axis or the immunity response. Though the drug delivers a number of side effects, it is considered to be less toxic than phenylbutazone, due to decreased rate of intestinal absorption.
Oxyphenbutazone is an NSAID that has been shown to preferentially kill non-replicating Mycobaterium tuberculosis maintained in media that simulates the mildly acidic, in vivo conditions where drug-resistant, non replicating subpopulations of the bacteria reside in hosts. The compound has little or no affect on replication M. tuberculosis grown in normal liquid cultures.
기타 정보
Air sensitive
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
I A Wasfi et al.
American journal of veterinary research, 58(6), 636-640 (1997-06-01)
To document disposition variables of phenylbutazone and its metabolite, oxyphenbutazone, in camels (Camelus dromedarius) after single i.v. bolus administration of phenylbutazone, with a view to making recommendation on avoiding violative residues in racing camels. 6 healthy camels (4 males, 2
A Kadir et al.
Journal of veterinary pharmacology and therapeutics, 20(1), 54-60 (1997-02-01)
Phenylbutazone was administered intravenously and intramuscularly at a dosage rate of 4.4 mg/kg to a group of 6 female camels in a two-period crossover study. After intravenous (i.v.) administration, disposition was characterised by a two-compartment open model, with a low
M D Veiga et al.
Journal of pharmaceutical and biomedical analysis, 28(5), 973-982 (2002-06-01)
The interactions between a nonsteroidal anti-inflammatory drugs, oxyphenbutazone (OPB), with two cyclodextrins, beta-cyclodextrin (beta-CD) and gamma-cyclodextrin (gamma-CD), have been studied in an aqueous medium and in the solid state. Differential scanning calorimetry, hot stage microscopy, thermogravimetric analysis and X-ray diffraction
Ulcer of the cecum during oxyphenbutazone (tandearil) therapy.
Debenham G P
Canadian Medical Association Journal, 94(22), 1182-1182 (1966)
B Razdan et al.
Drug development and industrial pharmacy, 25(9), 1051-1056 (1999-10-13)
Dissolution-dialysis studies of commercial tablets of oxyphenbutazone were carried out to establish the applicability of this technique for the in vitro evaluation of oxyphenbutazone dosage form. While disintegration time and dissolution rate studies did not give a true indication of
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.