360716
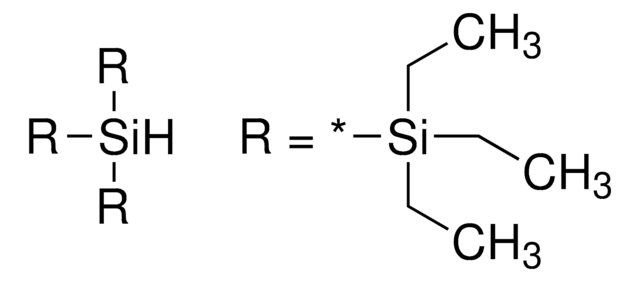
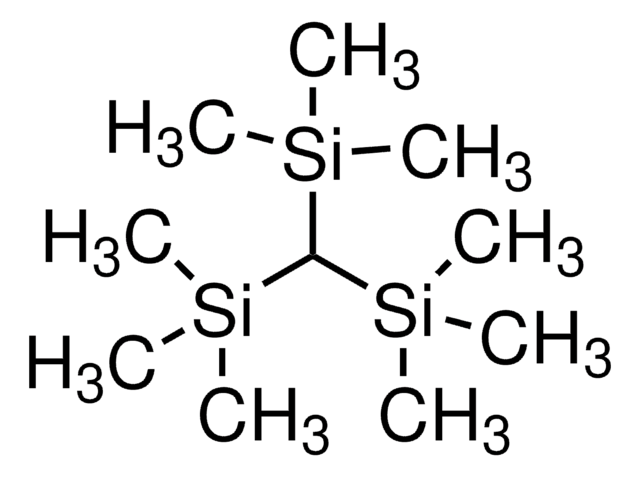
Tris(trimethylsilyl)silane
97%
Synonym(s):
1,1,1,3,3,3-Hexamethyl-2-trimethylsilyl-trisilane, TTMSS
About This Item
Recommended Products
assay
97%
form
liquid
reaction suitability
reagent type: reductant
refractive index
n20/D 1.489 (lit.)
bp
73 °C/5 mmHg (lit.)
density
0.806 g/mL at 25 °C (lit.)
SMILES string
C[Si](C)(C)[SiH]([Si](C)(C)C)[Si](C)(C)C
InChI
1S/C9H28Si4/c1-11(2,3)10(12(4,5)6)13(7,8)9/h10H,1-9H3
InChI key
SQMFULTZZQBFBM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Used in:
- Hydrosilylations
- Radical reactions
- Reductions of acid chlorides
- Reductions of carbon-halogen bonds
- Hydrosilations of carbonyls
- Another common application involves the use of the tris(trimethylsilyl)silyl (TTMSS, or super silyl) group when complexed with transition metals and main group elements
- More recently, the super silyl group is being utilized in carbon–carbon bond forming reactions
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Explore methods for molecular monolayers on silicon surfaces, their properties, and applications in molecular electronics and sensing.
Related Content
The Yamamoto group has employed several applications in catalysis.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)

![[4,4′-Bis(1,1-dimethylethyl)-2,2′-bipyridine] nickel (II) dichloride](/deepweb/assets/sigmaaldrich/product/structures/471/091/6faa29b1-bf8a-4d87-90b2-4cc55e082620/640/6faa29b1-bf8a-4d87-90b2-4cc55e082620.png)