One-step Feed Optimization for Upstream Cell Culture Processes
Optimizing media and feed formulations is essential for efficient and cost-effective upstream workflows. While there are many aspects to consider for optimizing formulations and cell culture processes, here we’ll discuss a one-step feed optimization using EX-CELL® Advanced CHO Feed 1 and Cellvento® 4Feed to improve monoclonal antibody (mAb) producing clones.
Why Use a Single Feed?
A single feed eliminates the need for extra manipulation and connections between bioreactors and media bottles or bags. This reduces the risk of contamination and simplifies the process.
About the EX-CELL® Advanced CHO Feed 1 and Cellvento® 4Feed
These feeds are part of our fed-batch media and feed platform systems. Both systems are chemically defined and suitable for a diverse set of CHO cell lines grown in suspension. EX-CELL® Advanced CHO Feed 1 contains a high concentration of key components and was developed using multivariate analysis and data mining to establish correlations between raw materials, and critical process and product attributes.
Cellvento® 4Feed is a highly concentrated feed (>130 grams per liter), which is reconstituted at neutral pH. Incorporation of modified amino acids and amino acid derivatives supports the slow release of amino acids such as cysteine and tyrosine, which is advantageous to the culture process. Additionally, modified amino acids in the feed design allow for reconstitution in a single preparation under neutral pH, eliminating the need to manage multiple feed additions prepared under different pH values and fed separately into the culture.
Optimizing EX-CELL® Advanced CHO Feed 1 and Cellvento® 4Feed Ratios for Greater Yields
EX-CELL® Advanced CHO Feed 1 and Cellvento® 4Feed can be combined and fed as a simple unit operation; each feed is hydrated individually and mixed as per the desired ratio. This one-step feed optimization solution using feed blends can deliver a twofold increase in titers for some production clones.
We examined the viable cell density and IgG titers from a one-step feed optimization for three clones from an IgG cell line development project (designated mAb01) generated using the CHOZN® GS CHO cell line development platform with UCOE® expression technology (CHOZN® & UCOE® combined platform). In addition to the individual EX-CELL® Advanced Feed 1 and Cellvento® 4Feed formulations, a single feed blend (50:50) was evaluated which consisted of 50 percent of each feed. Clone A preferred the 50:50 feed blend as evidenced by the higher viable cell densities and titers, whereas clones B and C had similar performance in all three conditions (Figure 1).
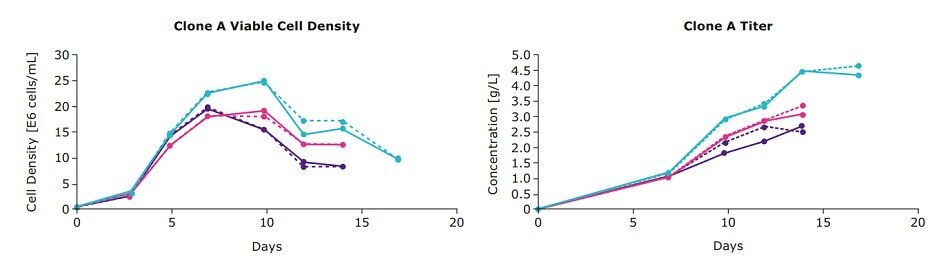
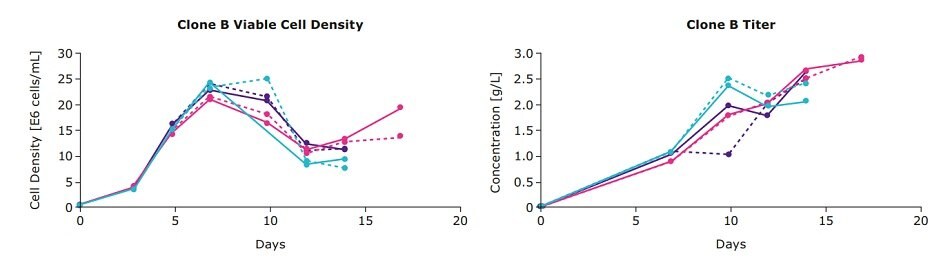

Figure 1.One-step feed optimization for mAb01 fed-batch cultures in spin tubes.
Clone A and B were subsequently scaled to 3 L and 50 L Mobius® bioreactors using the 50:50 feed blend and showed comparable results to the spin tubes data, demonstrating the scalability of the feed blend (Figure 2).

Figure 2.Viable cell density and titers from mAb01 clones A and B in 3 L and 50 L Mobius® bioreactors.
Figure 3 summarizes results from the one-step feed optimization for three clones from another IgG cell line development project (mAb03) generated with the CHOZN® & UCOE® combined platform using three feed blends. The clones preferred different feed blends and the mechanism of performance improvement varied from clone to clone. For example, clone D preferred the individual Cellvento® 4Feed whereas clone E preferred the 50:50 feed blend; clone F preferred both the 50:50 and the blend with 75 percent Cellvento® 4Feed (75:25). In terms of mechanisms, clone D and clone E showed improvement in viable cell density leading to higher titers. For clone F, improvement was observed in specific productivity.

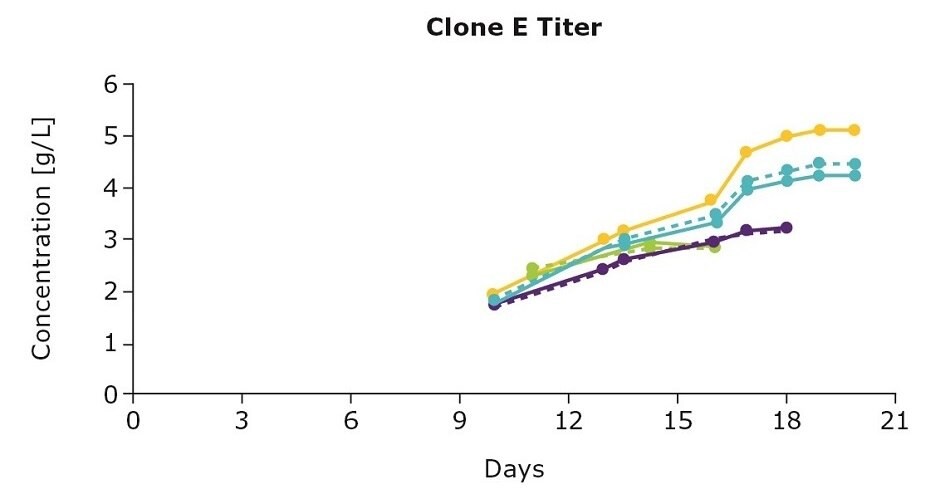

Figure 3.One-step feed optimization for mAb03 fed-batch cultures in spin tubes.
A similar scale-up experiment was performed for mAb03 to demonstrate scalability of the individual feed, Cellvento® 4Feed (Figure 4). The peak viable cell density and titer of the three clones in the 3 L Mobius® bioreactors was similar to that observed in the spin tubes screening process.

Figure 4.Viable cell density and titers from clones of mAb03 in 3 L Mobius® bioreactors.
One-step Feed Optimization for Difficult to Express Proteins
The one-step feed optimization process can also be used with difficult to express proteins such as bispecific antibodies. Screening results, for clones generated with the CHOZN® & UCOE® combined platform, with two feed blends and the individual feeds along with subsequent scale-up for three clones with the best performing feed blend (67:33) are shown in Figures 5 and 6, respectively. These clones required different feed blends to achieve the highest titer (Figure 5) and the process was scalable (Figure 6). Titers of 2 to 3.5 g/L for the bispecific antibody were achieved in a 3 L Mobius® bioreactor using the best performing feed screened during the one-step feed optimization.

Figure 5.One-step feed optimization for bispecific antibody fed-batch cultures in spin tubes.

Figure 6.Viable cell density and titers of bispecific antibody clones in 3 L Mobius® bioreactors.
Composition of Feed Blends
To further explore the differences between feed blends, we developed a heat map based on the main nutrient components in the feed blends. The components were grouped as amino acids, vitamins, lipids, trace metals, and other components along with a total concentration classification (Figure 7). The different feed blends offer a diversity in nutrient components, where some blends may be lower in certain nutrients but higher in others. Given this diversity, it is advisable to screen all the feed blend ratios to increase the coverage and probability of finding a feed that is preferred by the clone for optimized performance.

Figure 7.Heatmap describing EX-CELL® Advanced, Cellvento® 4Feed and their blends.
Conclusion
Platform feed screens using feed blends accelerate and simplify media and process development by eliminating the need for multiple feeds and inputs into the bioreactor. This simple methodology makes it easier to screen multiple feed blends to find one that provides the best viable cell density and titer.
To continue reading please sign in or create an account.
Don't Have An Account?