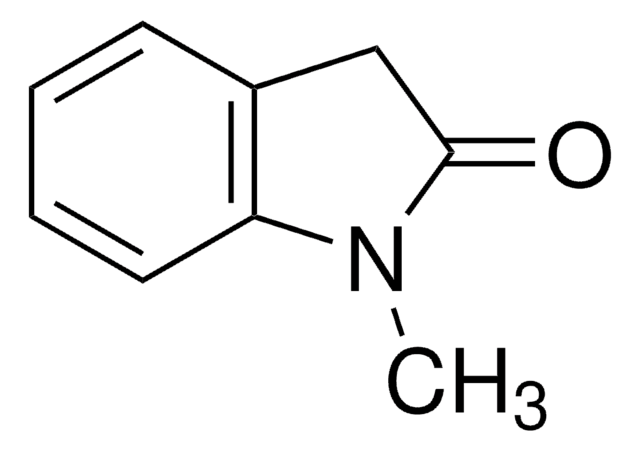
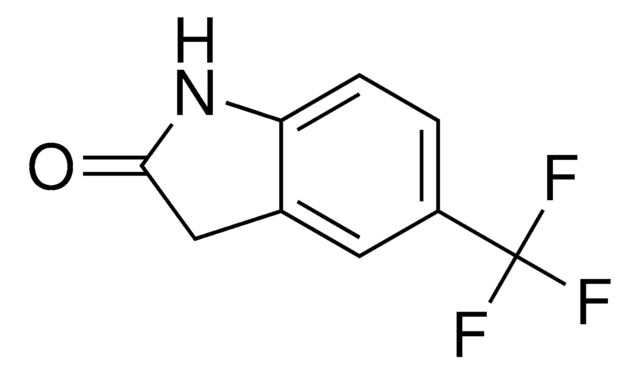
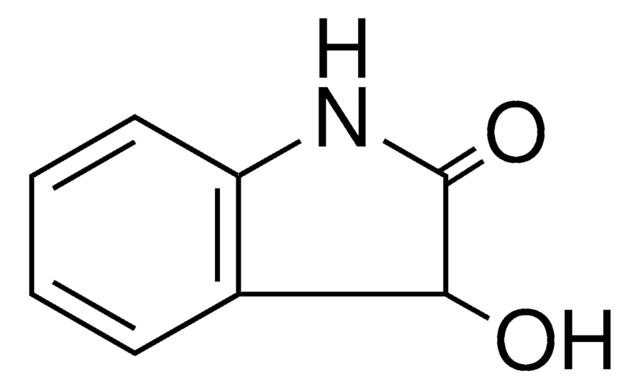
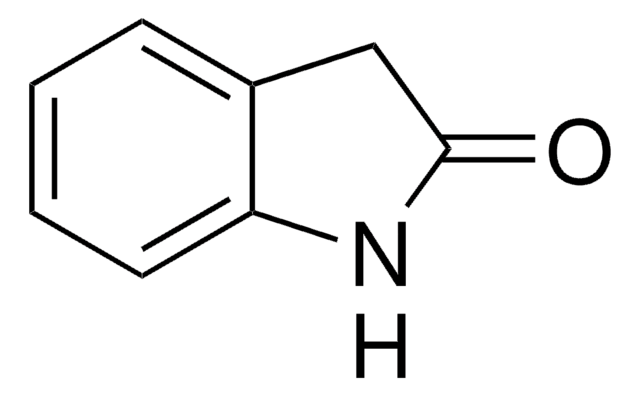
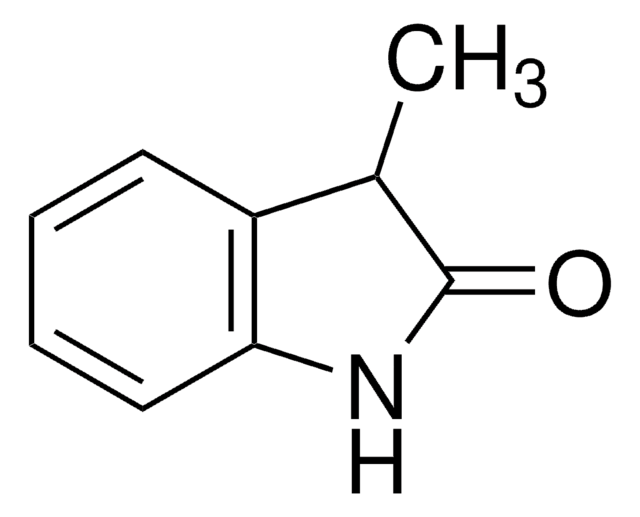
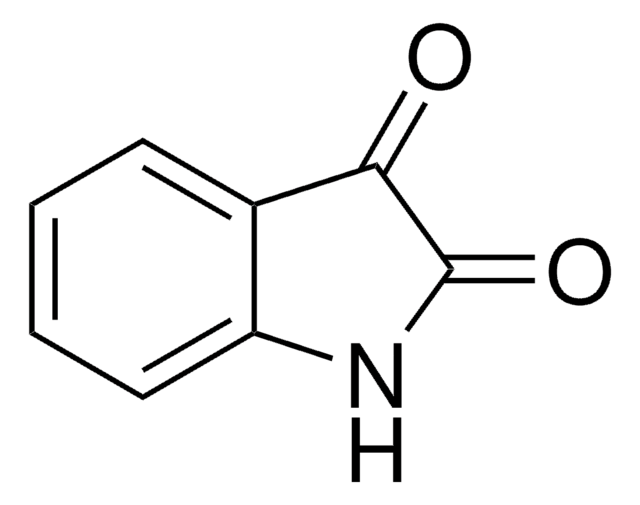
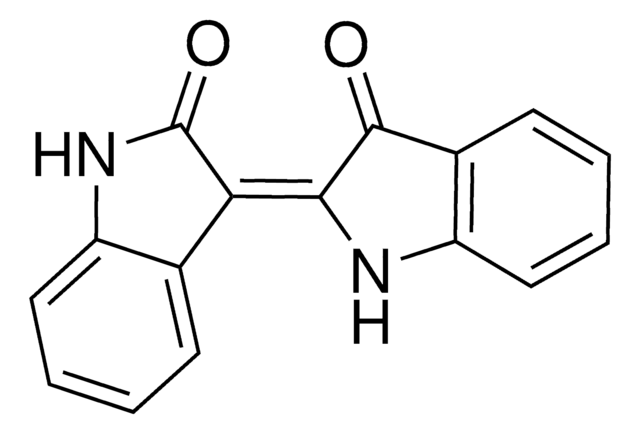
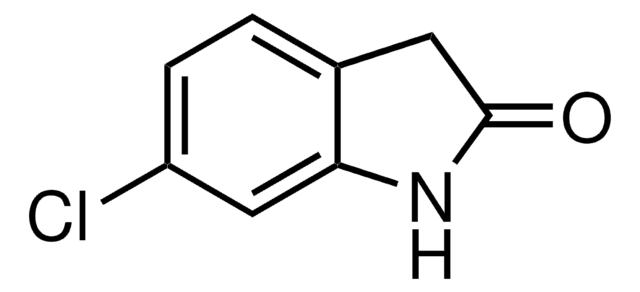
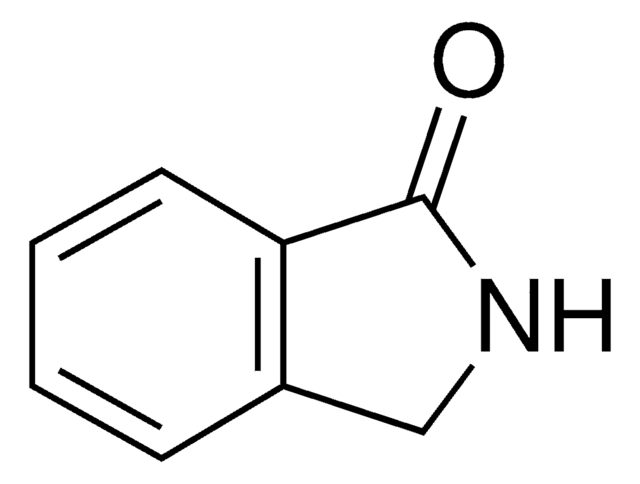
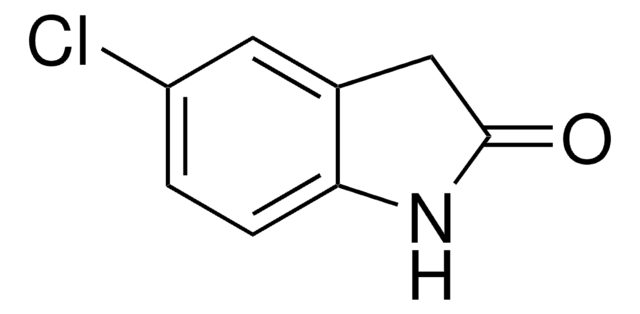
おすすめの製品
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
O9808-25G:
O9808-100G:
O9808-BULK:
O9808-VAR:
O9808-5G:
この製品を見ている人はこちらもチェック
Rahul R Khanwelkar et al.
Bioorganic & medicinal chemistry, 18(13), 4674-4686 (2010-06-24)
A series of new ureidoindolin-2-one derivatives were synthesized and evaluated as inhibitors of receptor tyrosine kinases. Investigation of structure-activity relationships at positions 5, 6, and 7 of the oxindole skeleton led to the identification of 6-ureido-substituted 3-pyrrolemethylidene-2-oxindole derivatives that potently
C Lanzi et al.
International journal of cancer, 85(3), 384-390 (2000-02-01)
ret-derived oncogenes are frequently and specifically expressed in thyroid tumors. In contrast to the ret receptor, ret oncoproteins are characterized by ligand-independent tyrosine-kinase activity and tyrosine phosphorylation. In this study, novel synthetic arylidene 2-indolinone compounds were evaluated as inhibitors of
Morteza Bararjanian et al.
The Journal of organic chemistry, 75(9), 2806-2812 (2010-04-15)
An efficient palladium-catalyzed protocol for the synthesis of 3-arylidene-2-oxindoles has been developed. In this approach, a sequential one-pot six-component reaction via Ugi/Heck carbocyclization/Sonogashira/nucleophilic addition was used for the synthesis of the desired skeleton.
Fangrui Zhong et al.
Organic letters, 13(1), 82-85 (2010-12-09)
The first tertiary amine catalyzed enantioselective Morita-Baylis-Hillman (MBH) reaction of isatins with acrylates has been demonstrated, allowing asymmetric synthesis of biologically significant 3-substituted-3-hydroxy-2-oxindoles in good yields and with excellent enantioselectivities. The C6'-OH group of β-isocupreidine (β-ICD) is believed to facilitate
Xianjie Chen et al.
Organic letters, 15(8), 1846-1849 (2013-04-09)
An asymmetric organo- and gold-catalyzed one-pot sequential Mannich/hydroamination reaction has been developed. Using this protocol, spiro[pyrrolidin-3,2'-oxindole] derivatives were synthesized in good yields (up to 91%) and excellent enantioselectivities (up to 97% ee).
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)