すべての画像(2)
About This Item
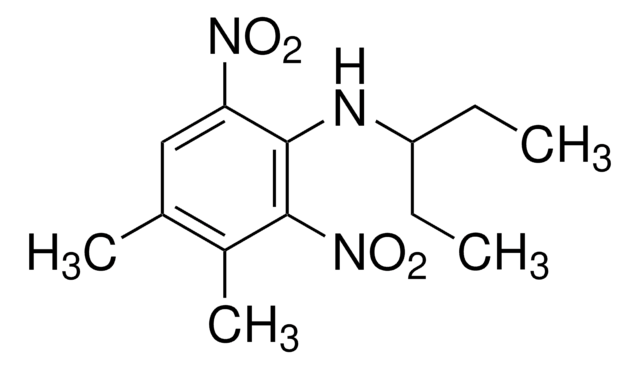
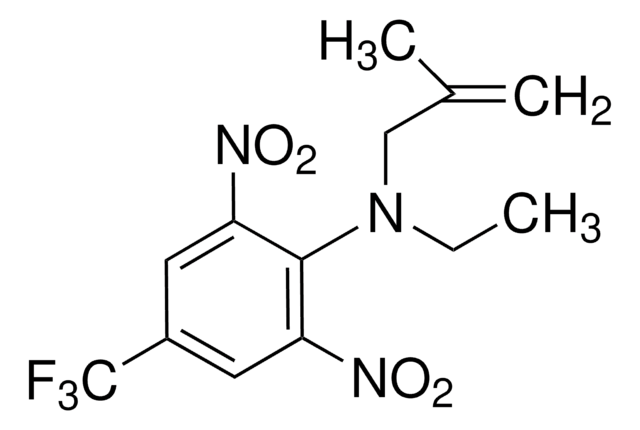
実験式(ヒル表記法):
C14H16F3N3O4
CAS番号:
分子量:
347.29
Beilstein:
2179006
EC Number:
MDL番号:
UNSPSCコード:
41116107
PubChem Substance ID:
NACRES:
NA.24
おすすめの製品
グレード
analytical standard
品質水準
製品種目
PESTANAL®
シェルフライフ
limited shelf life, expiry date on the label
テクニック
HPLC: suitable
gas chromatography (GC): suitable
アプリケーション
agriculture
environmental
フォーマット
neat
SMILES記法
CCCN(CC1CC1)c2c(cc(cc2[N+]([O-])=O)C(F)(F)F)[N+]([O-])=O
InChI
1S/C14H16F3N3O4/c1-2-5-18(8-9-3-4-9)13-11(19(21)22)6-10(14(15,16)17)7-12(13)20(23)24/h6-7,9H,2-5,8H2,1H3
InChI Key
ITVQAKZNYJEWKS-UHFFFAOYSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
アプリケーション
Refer to the product′s Certificate of Analysis for more information on a suitable instrument technique. Contact Technical Service for further support.
おすすめ製品
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
法的情報
PESTANAL is a registered trademark of Merck KGaA, Darmstadt, Germany
適切な製品が見つかりませんか。
製品選択ツール.をお試しください
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2
保管分類コード
11 - Combustible Solids
WGK
WGK 2
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
dust mask type N95 (US), Eyeshields, Gloves
最新バージョンのいずれかを選択してください:
A R Jacobson et al.
Chemical research in toxicology, 1(5), 304-311 (1988-09-01)
Fluorine NMR spectroscopy has been used to identify and quantitate metabolites of profluralin appearing in the urine of rats postingestion of the herbicide. The chemical shift of the trifluoromethyl group was shown to be a useful indicator of the chemical
N D Camper et al.
Journal of environmental science and health. Part. B, Pesticides, food contaminants, and agricultural wastes, 15(5), 457-473 (1980-01-01)
The degradation of profluralin [N-(cyclopropylmethyl)-alpha,alpha,alpha-trifluoro-2,6-dinitro-N-propyl-p-toluidine] and trifluralin (alpha,alpha,alpha-trifluoro-2,6-dinitro-N,N-dipropyl-p-toluidine) was studied under aerobic and anaerobic soil conditions. Three soils (Goldsboro loamy sand, Cecil loamy sand, Drummer clay loam) were each treated with 1 ppmw herbicide; anaerobic conditions were maintained by flooding.
R C Honeycutt et al.
Journal of environmental pathology and toxicology, 3(5-6), 21-33 (1980-06-01)
Recent developments in the technology of removal and characterization of bound residues of two herbicides, nitrofen and profluralin, from plants and soil will be reviewed. 14C-Nitrofen was found to be metabolized into starch in wheat grain as well as bound
M J Arrowood et al.
FEMS microbiology letters, 136(3), 245-249 (1996-03-01)
Despite the evaluation of over 100 antimicrobial drugs, the diarrheal disease cryptosporidiosis has remained refractory to treatment. We report the evaluation of five dinitroaniline herbicides including trifluralin, profluralin, nitralin, pendimethalin, and fluchloralin for anticryptosporidial activity in an in vitro cultivation
T R Edgerton et al.
Journal of analytical toxicology, 9(1), 15-19 (1985-01-01)
A method is presented for the analysis of trace amounts of dinitroaniline herbicides in tissue and excreta. The method employs extraction of the tissue or excreta with organic solvent, clean up by liquid/liquid partitioning or silica gel chromatography, and ultimate
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)