추천 제품
Quality Level
분석
95%
반응 적합성
reagent type: ligand
reaction type: [1,2]-Wittig Rearrangement
refractive index
n20/D 1.466 (lit.)
bp
42-43 °C/10 mmHg (lit.)
density
1.187 g/mL at 25 °C (lit.)
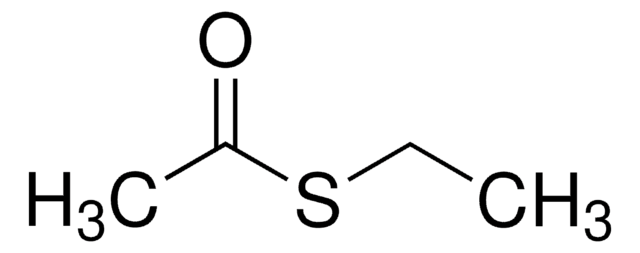
작용기
ester
thiol
SMILES string
COC(=O)CS
InChI
1S/C3H6O2S/c1-5-3(4)2-6/h6H,2H2,1H3
InChI key
MKIJJIMOAABWGF-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Methyl thioglycolate reacts with nonprotein component of the antitumor antibiotic neocarzinostatin to form 1:1 adduct. It reacts with isothiocyanate to form Rhodanine.
애플리케이션
Methyl thioglycolate was used in the preparation of:
A General Strategy for Organocatalytic Activation of C–H Bonds via Photoredox Catalysis: Direct Arylation of Benzylic Ethers
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
- 3-carbomethoxy-4- oxotetrahydrothiopyran, 2- and 4-carbomethoxy-3-oxotetrahydrothiophene
- methyl thioglycolate and aminoethanethiol conjugated gold nanorods.
A General Strategy for Organocatalytic Activation of C–H Bonds via Photoredox Catalysis: Direct Arylation of Benzylic Ethers
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Inhalation - Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point (°F)
132.8 °F - closed cup
Flash Point (°C)
56 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
T Kusakabe et al.
Biochemistry, 34(31), 9944-9950 (1995-08-08)
A double-stranded DNA cleavage mechanism by a novel enediyne type antitumor antibiotic, dynemicin A, has been investigated through sequence-dependent strand breakage of a series of duplexes containing a single nucleotide gap. We found that (1) dynemicin A breaks specifically at
Photodegradation of methyl thioglycolate particles as a proxy for organosulphur containing droplets.
Samantha Seng et al.
Physical chemistry chemical physics : PCCP, 20(29), 19416-19423 (2018-06-28)
Understanding the formation and transformation of sulphur-rich particles is of prime importance since they contribute to the global atmospheric sulphur budget. In this work, we performed a series of experiments on a photoactive organosulphur compound namely, methyl thioglycolate, as a
Luca Bertini et al.
Dalton transactions (Cambridge, England : 2003), 40(30), 7704-7712 (2011-06-01)
Amavadin is an unusual octa-coordinated V(IV) complex isolated from Amanita muscaria mushrooms. The outer-sphere catalytic properties of such a complex toward several oxidation reactions are well known. Nevertheless, a remarkable example exists, in which the V(V) (d(0)) oxidized form of
Sumati Murli et al.
Applied and environmental microbiology, 71(8), 4503-4509 (2005-08-09)
Chemobiosynthesis (J. R. Jacobsen, C. R. Hutchinson, D. E. Cane, and C. Khosla, Science 277:367-369, 1997) is an important route for the production of polyketide analogues and has been used extensively for the production of analogues of 6-deoxyerythronolide B (6-dEB).
W A LaMarr et al.
Proceedings of the National Academy of Sciences of the United States of America, 95(1), 102-107 (1998-02-21)
DNA superhelical tension, an important feature of genomic organization, is known to affect the interactions of intercalating molecules with DNA. However, the effect of torsional tension on nonintercalative DNA-binding chemicals has received less attention. We demonstrate here that the enediyne
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.