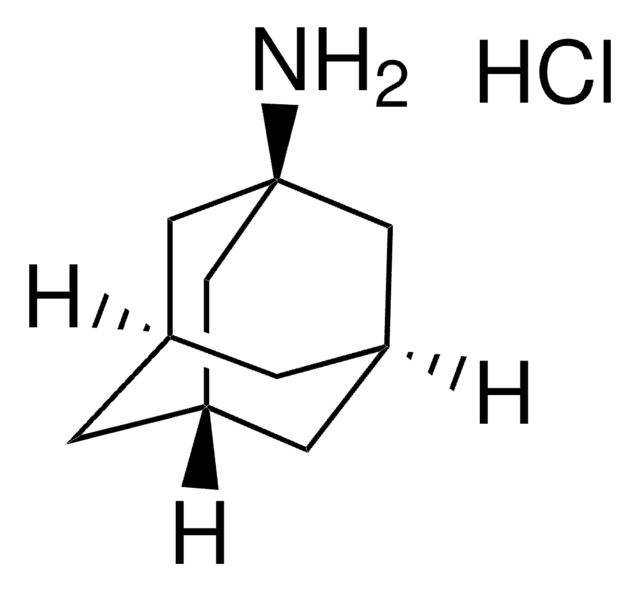
153818
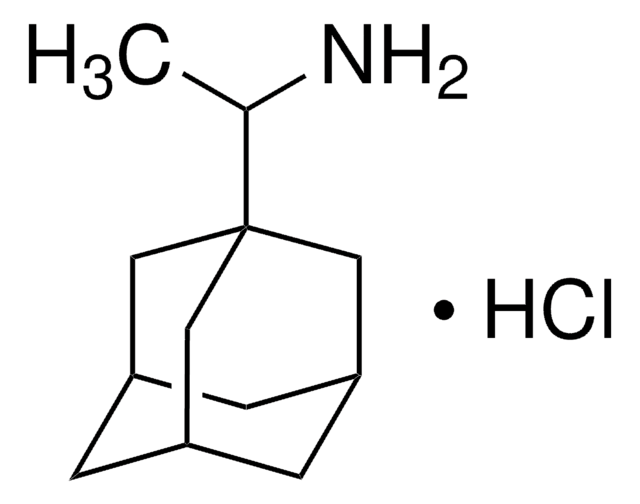
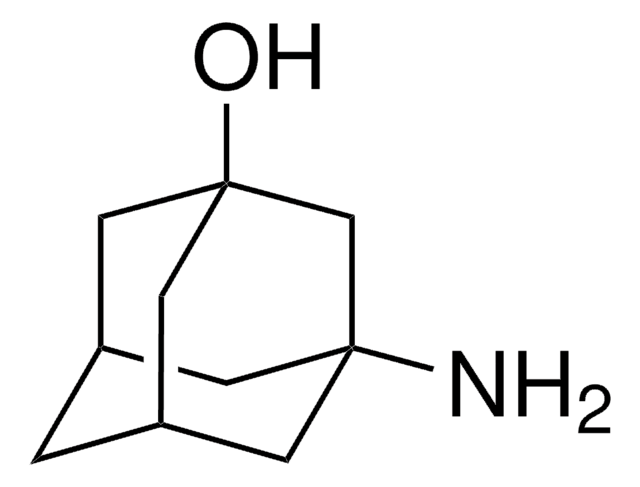
2-Adamantylamine hydrochloride
99%
동의어(들):
2-Adamantanamine hydrochloride, 2-Aminoadamantane hydrochloride
로그인조직 및 계약 가격 보기
모든 사진(2)
About This Item
실험식(Hill 표기법):
C10H17N · HCl
CAS Number:
Molecular Weight:
187.71
Beilstein:
4297901
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
양식:
solid
분석:
99%
추천 제품
일반 설명
The HPLC assay of 2-adamantylamine hydrochloride after pre-column derivatization with 4-fluoro-7-nitro-2,1,3-benzoxadiazole has been studied.
애플리케이션
2-Adamantylamine hydrochloride was used to prepare 2-adamantylamide of 2′-(carboxymethoxime)-olivomycin I.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Alina A Okhina et al.
Journal of pharmaceutical and biomedical analysis, 180, 113039-113039 (2019-12-23)
A method of quantitative determination of camphecene, a new anti-influenza agent, in rat blood plasma based on LC-MS/MS was developed, validated and used to study the distribution of the agent between blood cells and blood plasma. The method was validated
S V Krapivin et al.
Biulleten' eksperimental'noi biologii i meditsiny, 116(11), 515-518 (1993-11-01)
The action of the new stimulant bromantane on spectra power EEG on Fourier of sensorimotor cortex, dorsal hippocamp and lateral hypothalamus of left and right hemispheres of brain of rat in free behavior was investigated. Bromantane leads to decreases in
Yasuhiko Higashi et al.
Biomedical chromatography : BMC, 20(5), 423-428 (2005-09-15)
Simultaneous HPLC assay of 1-adamantanamine hydrochloride (amantadine) and its four related compounds [2-adamantanamine hydrochloride (2-ADA), 1-adamantanmethylamine (ADAMA), 1-(1-adamantyl)ethylamine hydrochloride (rimantadine) and 3,5-dimethyl-1-adamantanamine hydrochloride (memantine)] in phosphate-buffered saline (pH 7.4) after pre-column derivatization with 4-fluoro-7-nitro-2,1,3-benzoxadiazole (NBD-F) was developed. Phosphate-buffered saline samples
Anna N Tevyashova et al.
The Journal of antibiotics, 62(1), 37-41 (2009-01-10)
A novel way of chemical modification of the antibiotic olivomycin I at the 2'-keto group of the side chain of the aglycone moiety was developed. Reaction of olivomycin I with the carboxymethoxylamine hemihydrochloride gave the key intermediate, 2'-carboxymethoxime-olivomycin I, which
C Clark et al.
Immunopharmacology, 21(1), 41-50 (1991-01-01)
The present in vitro investigations on amantadine (AmTd) and its isomer 2-aminoadamantane (2-NH2-Adam), and the corresponding analogs, 1-nitroadamantane (1-NO2-Adam) and 2-nitroadamantane (2-NO2-Adam), were undertaken to gain information about molecular features that might have a dominant role in inhibiting T lymphocyte
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.



![1-Bicyclo[1.1.1]pentylamine hydrochloride](/deepweb/assets/sigmaaldrich/product/structures/287/052/55f4f60a-a9e0-4ea2-b1e8-5b3f6ce0ff21/640/55f4f60a-a9e0-4ea2-b1e8-5b3f6ce0ff21.png)