추천 제품
Quality Level
분석
98%
형태
solid
mp
75-77 °C (lit.)
solubility
acetone: soluble 1%, clear, yellow
작용기
aldehyde
nitro
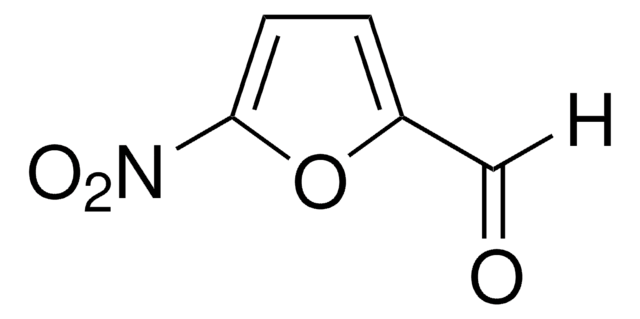
SMILES string
[H]C(=O)c1ccc(s1)[N+]([O-])=O
InChI
1S/C5H3NO3S/c7-3-4-1-2-5(10-4)6(8)9/h1-3H
InChI key
CHTSWZNXEKOLPM-UHFFFAOYSA-N
일반 설명
Diastereoselectivity in [4+2] cycloaddition of 1-methoxy-2-methyl-3-(trimethylsiloxy)-1,3-pentadiene with 5-nitro-2-thiophenecarboxaldehyde was investigated.
애플리케이션
5-Nitro-2-thiophenecarboxaldehyde was used in preparation of 2, 3-dihydro-2-(5-nitro-2-thienyl) quinazolin-4-(1H)-ones and various novel oxime ether derivatives, anti-protozoan agents.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Michael P Doyle et al.
Proceedings of the National Academy of Sciences of the United States of America, 101(15), 5391-5395 (2004-04-03)
Chiral dirhodium(II) carboxamidates are highly efficient catalysts for reactions between a variety of aldehydes and activated dienes. Catalyst loadings as low at 0.01 mol % have been realized with enantioselectivities up to 97%. Kinetic investigations reveal a pronounced electronic influence
Antibacterial 2,3-dihydro-2-(5-nitro-2-thienyl)-quinazolin-4(1H)-ones.
R J Alaimo et al.
Journal of medicinal chemistry, 15(3), 335-336 (1972-03-01)
Synthesis and< i> in vitro</i> anti-protozoan activity of new 5-nitrothiophene oxime ether derivatives.
Delmas F, et al.
European Journal of Medicinal Chemistry, 28(1), 23-27 (1993)
Ali Almasirad et al.
Iranian journal of pharmaceutical research : IJPR, 10(4), 727-731 (2011-10-01)
A series of new 2-(phenylthio) benzoylarylhydrazones were synthesized by acid-catalyzed condensation of hydrazide 3 with corresponding aldehydes. The chemical structures of the compounds were elucidated by FT-IR, (1)H-NMR and Mass spectra. All newly synthesized compounds were evaluated for their antimycobacterial
Jian Xu et al.
Food chemistry, 221, 1530-1538 (2016-12-17)
We synthesized a series of 4- or 5-functionalized TCT derivatives (1-12) and investigated their inhibitory activities and mechanisms on tyrosinase by using Spectrofluorimetry, 1H and 13C NMR titration and IR spectra. The results of the fluorescence spectra and NMR titrations
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.