추천 제품
분석
97%
mp
122-125 °C (lit.)
SMILES string
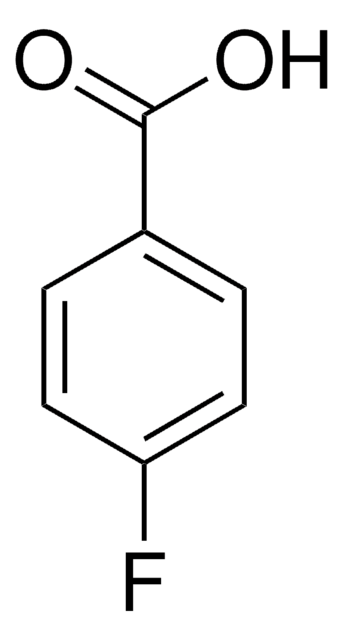
OC(=O)c1ccccc1F
InChI
1S/C7H5FO2/c8-6-4-2-1-3-5(6)7(9)10/h1-4H,(H,9,10)
InChI key
NSTREUWFTAOOKS-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
2-Fluorobenzoic acid may be employed in the preparation of zaragozic acid A analogs.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
S Y Lee et al.
Nuclear medicine and biology, 28(4), 391-395 (2001-06-08)
In vitro metabolism of acetylcholinesterase inhibitors containing 3-[(18)F]fluoromethylbenzyl- ([(18)F]1) and 4-[(18)F]fluorobenzyl-piperidine moieties ([(18)F]2) was studied and compared with the in vivo metabolism. Defluorination of the [(18)F]1 mainly occurred to generate [(18)F]fluoride ion both in vitro and in vivo. In contrast
Microbial degradation of synthetic organochlorine compounds.
K Motosugi et al.
Experientia, 39(11), 1214-1220 (1983-11-15)
J Toretsky et al.
Nuclear medicine and biology, 31(6), 747-752 (2004-07-13)
The clinical response to antitumor therapy is measured using imaging, such as CT or MRI, 6-12 weeks following chemotherapy treatment. The images at that time reflect both tumor cell death and new growth. Therefore, the amount of tumor cell death
U Schennen et al.
Journal of bacteriology, 161(1), 321-325 (1985-01-01)
Three strains of anaerobically benzoate-degrading, denitrifying bacteria of the genus Pseudomonas were able to grow on 2-fluorobenzoate as the sole carbon and energy source. Fluoride ion release was stoichiometric, and the reduction of dissolved organic carbon indicated total degradation. Cells
T S Chen et al.
The Journal of antibiotics, 47(11), 1290-1294 (1994-11-01)
Zaragozic acid A analogues are produced by an unidentified sterile fungus when it is exogenously supplied with 2-thiophenecarboxylic acid, 3-thiophenecarboxylic acid, 2-furoic acid, 2-fluorobenzoic acid, 3-fluorobenzoic acid, or 4-fluorobenzoic acid. The analogues carry 2-thiophenyl, 3-thiophenyl, 2-furyl, o-fluorophenyl, m-fluorophenyl, or p-fluorophenyl
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.