About This Item
추천 제품
양식
solid
Quality Level
광학 활성
[α]20/D +162°, c = 0.5 in benzene
반응 적합성
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
reaction type: Cross Couplings
mp
254-258 °C
작용기
phosphine
SMILES string
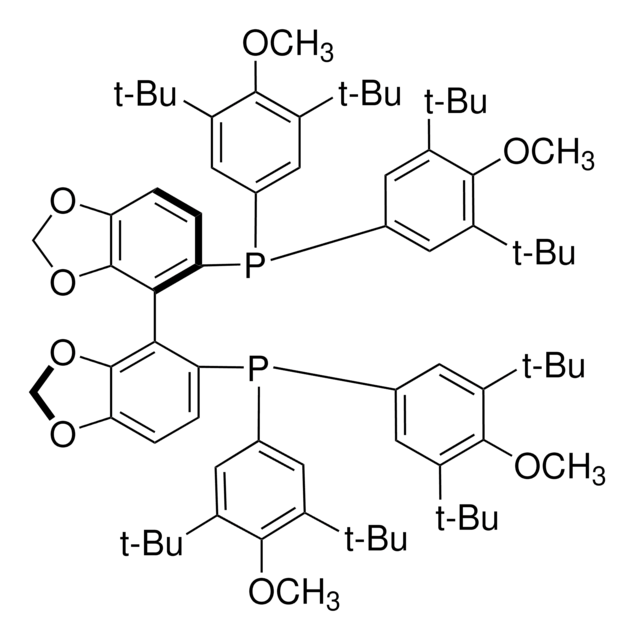
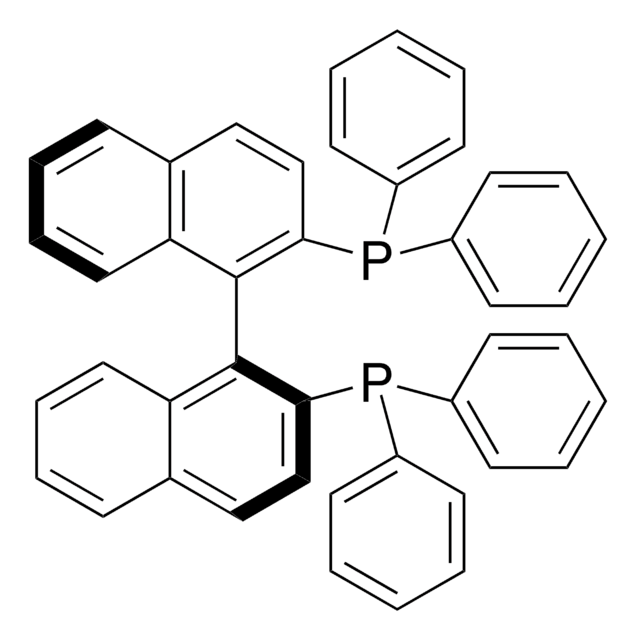
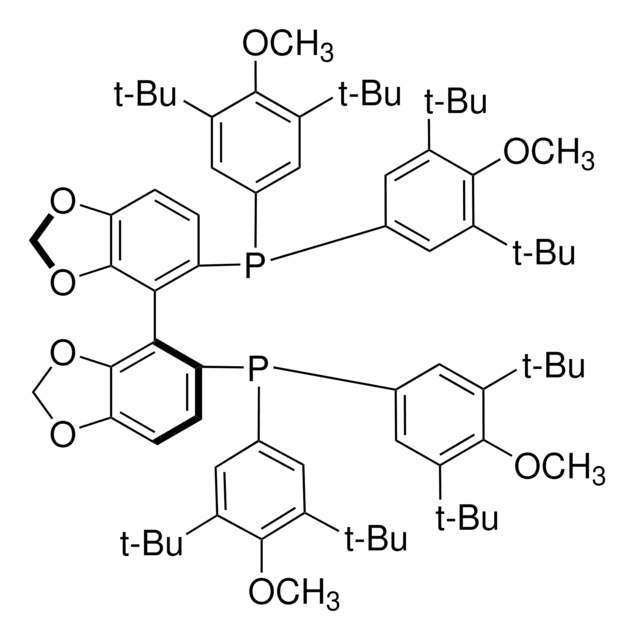
Cc1ccc(cc1)P(c2ccc(C)cc2)c3ccc4ccccc4c3-c5c(ccc6ccccc56)P(c7ccc(C)cc7)c8ccc(C)cc8
InChI
1S/C48H40P2/c1-33-13-23-39(24-14-33)49(40-25-15-34(2)16-26-40)45-31-21-37-9-5-7-11-43(37)47(45)48-44-12-8-6-10-38(44)22-32-46(48)50(41-27-17-35(3)18-28-41)42-29-19-36(4)20-30-42/h5-32H,1-4H3
InChI key
IOPQYDKQISFMJI-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
애플리케이션
Reactant serving as a precursor for:
- Catalysts used for reductive amination of ketones
- Rh(I)-catalyst for hydrogenation of acetamidoacrylic acid derivatives
- Chiral platinum catalysts for asymmetric Baeyer-Villiger oxidation of cyclic ketones
- CuI-Tol-BINAP catalysts for enantioselective Michael reactions of Grignard reagents to unsaturated esters
- BINAP Pt Dications for cation trapping
법적 정보
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
문서
The Baeyer-Villiger oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts the ketones to esters and the cyclic ketones to lactones.
We present an article concerning BINAP/SEGPHOS® Ligands and Complexes.
Hydrogenation, Asymmetric Catalysis, Binap, SEGPHOS®, Aldol reaction, Alkenylation, Arylation, Mannich reaction, Fluorination, Michael addition, Hydrosilylation, Cycloaddition, Takasago
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.