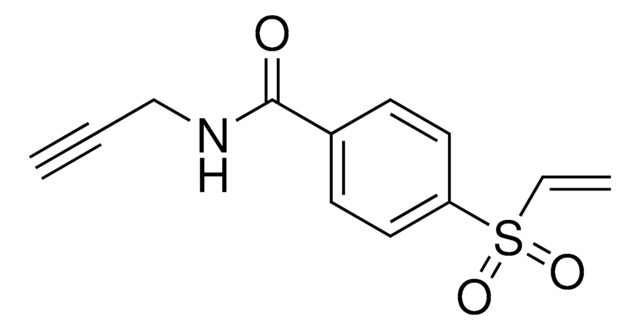
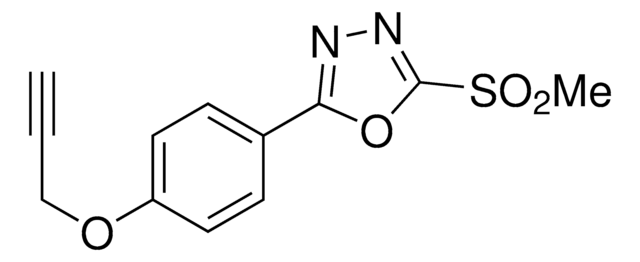
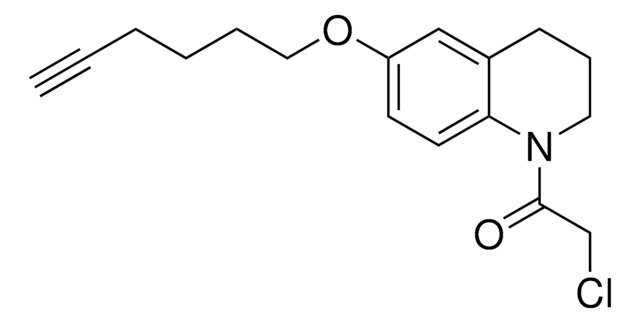
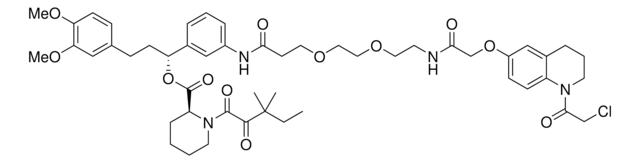
추천 제품
Quality Level
분석
≥95%
양식
powder
저장 온도
−20°C
SMILES string
CC1=CC(C(OI2C#C[Si](C)(C)C)=O)=C2C=C1
InChI
1S/C13H15IO2Si/c1-10-5-6-12-11(9-10)13(15)16-14(12)7-8-17(2,3)4/h5-6,9H,1-4H3
InChI key
MJEDSBVZRIKHTM-UHFFFAOYSA-N
애플리케이션
EBX2-alkyne is a hypervalent iodine reagent used for labeling cysteines. A method was developed using cysteine-reactive compounds including this one to allow for unbiased analysis of proteomic data in quantitative applications . The method uses light or heavy labeling with the isotopically labelled desthiobiotin azide (isoDTB) tag for mass spectrometry analysis . Analysis then uses the isotopic tandem orthogonal proteolysis activity-based protein profiling (isoTOP-ABPP) workflow .
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
Profiling the proteome-wide selectivity of diverse electrophiles.
Zanon P R A, et al.
ChemRxiv : the preprint server for chemistry (2021)
Eranthie Weerapana et al.
Nature, 468(7325), 790-795 (2010-11-19)
Cysteine is the most intrinsically nucleophilic amino acid in proteins, where its reactivity is tuned to perform diverse biochemical functions. The absence of a consensus sequence that defines functional cysteines in proteins has hindered their discovery and characterization. Here we
Keriann M Backus et al.
Nature, 534(7608), 570-574 (2016-06-17)
Small molecules are powerful tools for investigating protein function and can serve as leads for new therapeutics. Most human proteins, however, lack small-molecule ligands, and entire protein classes are considered 'undruggable'. Fragment-based ligand discovery can identify small-molecule probes for proteins
Patrick R A Zanon et al.
Angewandte Chemie (International ed. in English), 59(7), 2829-2836 (2019-11-30)
Rapid development of bacterial resistance has led to an urgent need to find new druggable targets for antibiotics. In this context, residue-specific chemoproteomic approaches enable proteome-wide identification of binding sites for covalent inhibitors. Described here are easily synthesized isotopically labeled
Eranthie Weerapana et al.
Nature, 468(7325), 790-795 (2010-11-19)
Cysteine is the most intrinsically nucleophilic amino acid in proteins, where its reactivity is tuned to perform diverse biochemical functions. The absence of a consensus sequence that defines functional cysteines in proteins has hindered their discovery and characterization. Here we
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.